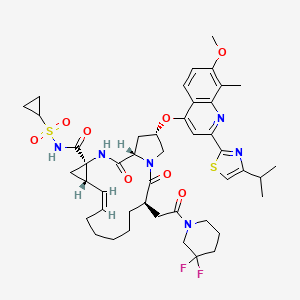
ACH-0142684, ACH-2684
HCV NS3 PR
USAN (YY-152) DELDEPREVIR
THERAPEUTIC CLAIM Treatment of Hepatitis C
CHEMICAL NAMES
1. Cyclopropa[e]pyrrolo[1,2-a][1,4]diazacyclopentadecine-14a(5H)-carboxamide, N-
(cyclopropylsulfonyl)-6-[2-(3,3-difluoro-1-piperidinyl)-2-oxoethyl]-
1,2,3,6,7,8,9,10,11,13a,14,15,16,16a-tetradecahydro-2-[[7-methoxy-8-methyl-2-[4-(1-
methylethyl)-2-thiazolyl]-4-quinolinyl]oxy]-5,16-dioxo-, (2R,6R,12Z,13aS,14aR,16aS)-
2. (2R,6R,12Z,13aS,14aR,16aS)-N-(cyclopropylsulfonyl)-6-[2-(3,3-difluoropiperidin-1-yl)-
2-oxoethyl]-2-({7-methoxy-8-methyl-2-[4-(1-methylethyl)thiazol-2-yl]quinolin-4-yl}oxy)-
5,16-dioxo-1,2,3,6,7,8,9,10,11,13a,14,15,16,16atetradecahydrocyclopropa[e]pyrrolo[1,2-a][1,4]diazacyclopentadecine-14a(5H)-
carboxamide
MOLECULAR FORMULA C45H56F2N6O8S2
MOLECULAR WEIGHT 911.1
SPONSOR Achillion Pharmaceuticals, Inc.
CODE DESIGNATION ACH-0142684, ACH-2684
CAS REGISTRY NUMBER 1229626-28-1
WHO NUMBER 9600
NOTE: This adoption statement replaces adoption N12/17 and the name neceprevir is hereby rescinded.
……………………………………………………………………………………………………….
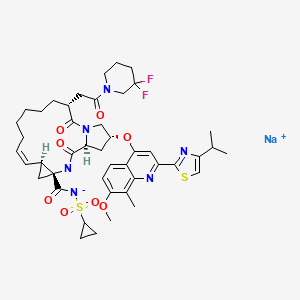
DELDEPREVIR SODIUM
USAN (yy-153) DELDEPREVIR SODIUM
THERAPEUTIC CLAIM Treatment of Hepatitis C
CHEMICAL NAMES
1. Cyclopropa[e]pyrrolo[1,2-a][1,4]diazacyclopentadecine-14a(5H)-carboxamide, N-
(cyclopropylsulfonyl)-6-[2-(3,3-difluoro-1-piperidinyl)-2-oxoethyl]-
1,2,3,6,7,8,9,10,11,13a,14,15,16,16a-tetradecahydro-2-[[7-methoxy-8-methyl-2-[4-(1-
methylethyl)-2-thiazolyl]-4-quinolinyl]oxy]-5,16-dioxo-, sodium salt (1:1),
(2R,6R,12Z,13aS,14aR,16aS)-
2. Sodium (cyclopropylsulfonyl){[(2R,6R,12Z,13aS,14aR,16aS)-6-[2-(3,3-difluoropiperidin-
1-yl)-2-oxoethyl]-2-({7-methoxy-8-methyl-2-[4-(1-methylethyl)thiazol-2-yl]quinolin-4-
yl}oxy)-5,16-dioxo-1,2,3,6,7,8,9,10,11,13a,14,15,16,16a-
tetradecahydrocyclopropa[e]pyrrolo[1,2-a][1,4]diazacyclopentadecin-14a(5H)-
yl]formyl]azanide
MOLECULAR FORMULA C45H55F2N6NaO8S2
MOLECULAR WEIGHT 933.1
SPONSOR Achillion Pharmaceuticals, Inc.
CODE DESIGNATION ACH-0142684.Na, ACH-2684.Na
CAS REGISTRY NUMBER 1298053-61-8
NOTE: This adoption statement replaces adoption N12/18 and the name neceprevir sodium
is hereby rescinded.
ACH-2684 is a HCV NS3 protease inhibitor in phase I clinical development at Achillion for the oral treatment of chronic hepatitis C genotype 1 and 3.
| WO 2010068761 | ||
| US 2010152103 |

COMPD 133
(2R,6R,14aR,16aS,Z)- N-(cyclopropylsulfonyl)- 6-(2-(3,3-difluoropiperidin- 1-yl)-2-oxoethyl)-2- (2-(2-isopropylthiazol- 4-yl)-7-methoxy-8- methylquinolin-4- yloxy)-5,16-dioxo- 1,2,3,5,6,7,8,9,10,11, 13a,14,14a,15,16,16a- hexadecahydrocyclopropa [e]pyrrolo[1,2- a][1,4] diazacyclopentadecine- 14a-carboxamide