DOI: 10.1039/C7GC03425C, Paper
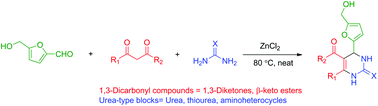
The use of the renewable platform molecule 5-hydroxymethylfurfural (HMF) in the multi-component Biginelli reaction has been investigated.
HMF in multicomponent reactions: utilization of 5-hydroxymethylfurfural (HMF) in the Biginelli reaction
Abstract
The use of the renewable platform molecule 5-hydroxymethylfurfural (HMF) in the multi-component Biginelli reaction has been investigated. Multicomponent reactions (MCR) using HMF offer straightforward access to novel fine chemicals. However, the peculiar reactivity and lower stability of HMF have limited its use in such strategies. In this paper, we report our results on the use of HMF in 3-component Biginelli reactions, leading in one single step to a series of functionalized dihydropyrimidinones obtained in moderate to good yields, with a broad substrate scope of 1,3-dicarbonyl compounds and urea building blocks. This is the first report on the use of HMF in this reaction. The CH2OH motif found in HMF provides useful functionalization for the target molecules, which cannot be offered by simpler aldehydes such as furfural.
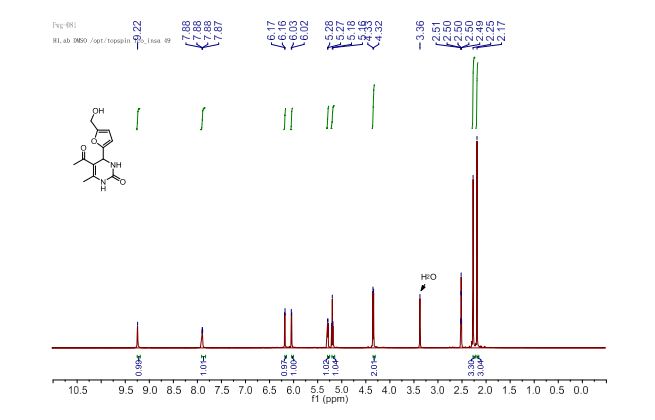
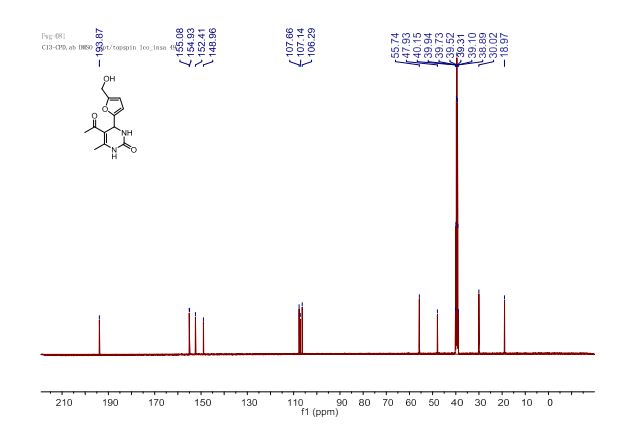
5-Acetyl-4-[5’-(hydroxymethyl)furan-2’-yl]-6-methyl-3,4-dihydropyrimidin-2(1H)-one (4a):
Reaction time: 8 h; Global yield: 86%; (78% yield after simple filtration + additional 8% yield after purification of the filtrate by column chromatography).
1H NMR (400 MHz, DMSO-d6) δ 9.22 (d, 1H, J = 1.2 Hz, H1), 7.88 (dd, 1H, J = 3.4, 1.2 Hz, H3), 6.16 (d, 1H, J = 3.1 Hz, H4’), 6.03 (d, 1H, J = 3.1 Hz, H3’), 5.27 (d, 1H, J = 3.4 Hz, H4), 5.18 (t, 1H, J = 5.6 Hz, OH), 4.33 (d, 2H, J = 5.6 Hz, CH2), 2.25 (s, 3H, CH3-C6), 2.17 (s, 3H, CH3CO).
13C NMR (100 MHz, DMSO-d6) δ 193.9 (COCH3), 155.1, 154.9 (C2’, C5’), 152.4 (C2), 149.0 (C6), 107.7 (C4’), 107.1 (C5), 106.3 (C3’), 55.7 (CH2OH), 47.9 (C4), 30.0 (CH3CO), 19.0 (CH3-C6).
HRMS (ESI) m/z: Calcd for [M+Na]+ C12H14N2NaO4 273.0846; Found 273.0850.
Research experience

E-mail: florence.popowycz@insa-lyon.fr

Yves QUENEAU
ICBMS INSA Lyon University of Lyon

E-mail: yves.queneau@insa-lyon.fr,













 .
. Corresponding author email mh_1395@yahoo.com
Corresponding author email mh_1395@yahoo.com