
US 8,007,830, US 5,565,473*PED, MERCK
MK-0476 (Montelukast, L-706631)
| NDA 021409 | APPR Jul 26, 2002 |
MONTELUKAST (Singulair® Oral Granules) helps to reduce asthma symptoms (coughing, wheezing, shortness of breath, or chest tightness) and control your asthma. It does not provide instant relief and cannot be used to treat a sudden asthma attack. It works only when used on a regular basis to help reduce inflammation and prevent asthma attacks. This drug is also helpful in improving seasonal allergies, like hay fever. Montelukast is effective in adults and children
Amongst the US approvals, tentative FDA approvals have been identified for generic Montelukast sodium, awarded to Endo, Glenmark, Mylan, Roxane, Sandoz, Teva and Torrent. The large number of generic authorisations awaiting launch in the UK is indicative of the likely competition the Singulair product will face across Europe upon SPC expiry
EP Pat. No. 480,717 discloses Montelukast sodium along with other related compounds and the methods for their preparation. The reported method of synthesis proceeds through corresponding methyl ester namely, Methyl 2-[(3S)-[3-[(2E)-(7-chloroquinolin – 2yl) ethenyl] phenyl] – 3 – hydroxypropyl] benzoate and involves coupling methyl 1- (mercaptomethyi) cyclopropaneacetate with a mesylate generated in-situ. The methyl ester of Montelukast is hydrolyzed to free acids and the latter converted directly to Montelukast Sodium salt (Scheme -1). The process is not particularly suitable for large – scale production because it requires tedious chromatographic purification of the methyl ester intermediate and / or the final product and the product yield is low.
Scheme -1
U.S. Pat. No. 5,614632 disclosed a process for the preparation of crystalline Montelukast sodium, which comprises of the following steps (Scheme – 2):
■ Reaction of methyl 2-[3(S)-[3-[2-(7-chloroquinolin -2-yl) ethenyl] phenyl] -3- hydroxypropyl benzoate (I) with Grignard reagent, methyl magnesium chloride in presence of cerium chloride to give Diol (II) ■ Reaction of Diol (II) with methane sulfonyl chloride to afford 2-[2-[3 (s)-[3- (2-(7-chloro quinolin-2yl) ethenyl] phenyl]- 3 – methane sulfonyloxy propyl] phenyl] -2-propanol (III)
■ Condensation of 2-[2-[3(s)-[3-(2-(7-chloro quinolin – 2-yl) ethenyl] phenyl] –
3 – methane sulfonyloxypropyl] phenyl] – 2- propanol (III) with dilithium anion of 1-mercaptomethyl) cyclopropaneacetic acid, which has been generated by the reaction of l-(mercaptomethyl)cyclopropaneacetic acid (IV)with n-Butyl lithium
■ Isolation of the condensed product, Montelukast as solid Montelukast dicyclohexylamine salt
■ Purification and conversion of Montelukast dicyclohexylamine salt into Montelukast sodium
■ Crystallization of Montelukast sodium from a mixture of toluene and acetonitrile
The process disclosed in U.S Pat. No. 5,614,632 further involved the reaction of Diol (II) with methane sulfonyl chloride involves the reaction temperature of about – 25°C and the storage condition of the intermediate, 2-[2-[3(s)-[3-(2-(7-chloro quinolin – 2-yl) ethenyl] pheny] -3 -methane sulfonyloxy propyl] phenyl] -2-propanol (III) at temperature below – 150C for having the stability. The process further involves the reaction, formation of dilithium anion of l-(mercaptomethyl) cyclopropaneacetic acid which requires the usage of n-Butyl lithium, a highly flammable and hazardous reagent and the reaction is at temperature below -5°C further requires anhydrous conditions. Scheme – 2

Montelukast (trade names Singulair, Montelo-10, and Monteflo and Lukotas in India) is a leukotriene receptor antagonist(LTRA) used for the maintenance treatment of asthma and to relieve symptoms of seasonal allergies.[1][2] It is usually administered orally once a day. Montelukast is a CysLT1 antagonist; it blocks the action of leukotriene D4 (and secondary ligands LTC4 and LTE4) on the cysteinyl leukotriene receptor CysLT1 in the lungs and bronchial tubes by binding to it. This reduces the bronchoconstriction otherwise caused by the leukotriene and results in less inflammation.
Because of its method of operation, it is not useful for the treatment of acute asthma attacks. Again because of its very specificmechanism of action, it does not interact with other asthma medications such as theophylline.
Another leukotriene receptor antagonist is zafirlukast (Accolate), taken twice daily. Zileuton (Zyflo), an asthma drug taken four times per day, blocks leukotriene synthesis by inhibiting 5-lipoxygenase, an enzyme of the eicosanoid synthesis pathway.
The Mont in Montelukast stands for Montreal, the place where Merck developed the drug.[3]
Singulair was covered by U.S. Patent No. 5,565,473[9] which expired on August 3, 2012.[10] The same day, the FDA approved several generic versions of montelukast.[11]
On May 28, 2009, the United States Patent and Trademark Office announced their decision to launch a reexamination of the patent covering Singulair. The decision to reexamine was driven by the discovery of references that were not included in the original patent application process. The references were submitted through Article One Partners, an online research community focused on finding literature relating to existing patents. The references included a scientific article produced by a Merck employee around the key ingredient of Singulair, and a previously filed patent in the same technology area.[12]
On December 17, 2009, the U.S. Patent and Trademark Office determined that the patent in question was valid based on the initial reexamination and new information provided.[13]
Montelukast is currently available in film coated tablet and orodispersible tablet formulations for once-daily administration, and also available as an oral granule formulation which is specifically designed for administration to paediatric patients.
Patent family US17493193A claims crystalline Montelukast sodium and processes for its preparation . Patents within this family are not considered to be a constraint to generic competition because the protected technology may possibly be circumvented by the synthesis and use of different molecular forms and/or salts. Patent family US33954901P relates to the specific marketed oral granule formulation of Montelukast.
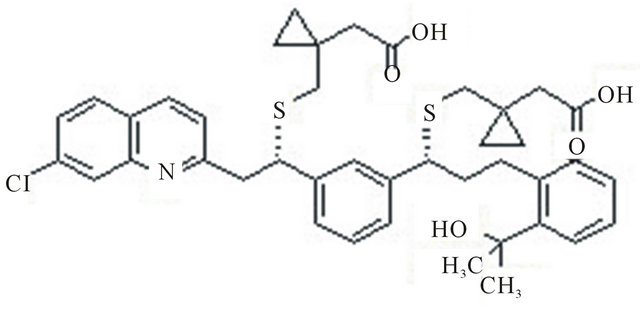
The chemical name of montelukast sodium is: Sodium 1-[[[(1R)-1-[3-[(1E)-2-(7-chloro-2-quinolinyl)ethenyl]phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]thio]methyl]cyclopropaneacetic acid and its structure is represented as follows:
-
Montelukast is apparently a selective, orally active leukotriene receptor antagonist that inhibits the cysteinyl leukotriene CysLT1 receptor.
-
U.S. patent No. 5,565,473 (“‘473 patent”) is listed in the FDA’s Orange Book for montelukast sodium. The ‘473 patent recites a broad class of leulcotriene antagonists as “anti-asthmatic, anti-allergic, anti-inflammatory, and cycloprotective agents” represented by a generic chemical formula. ‘473 patent, col. 2,1. 3 to col. 4,1. 4. Montelukast is among the many compounds represented by that formula. The ‘473 patent also refers to pharmaceutical compositions of the class of leukotriene antagonists of that formula with pharmaceutically acceptable carriers. Id. at col. 10,11. 42-46.
-
Montelukast sodium is currently marketed by Merck in the form of film coated tablets and chewable tablets under the trade name Singular®. The film coated tablets reportedly contain montelukast sodium and the following inactive ingredients: microcrystalline cellulose, lactose monohydrate, croscarmellose sodium, hydroxypropylcellulose, magnesium stearate, titanium dioxide, red ferric oxide, yellow ferric oxide, and carnauba wax. The chewable tablets reportedly containmontelukast sodium and the following inactive ingredients: mannitol, microcrystalline cellulose, hydroxypropylcellulose, red ferric oxide, croscarmellose sodium, cherry flavor, aspartame, and magnesium stearate. Physicians’ Desk Reference, 59th ed. (2005), p. 2141.
-
However, there is a need in the art to improve the stability of compositions of montelukast and particularly those of the sodium salt.
Montelukast sodium is a leukotriene antagonist and inhibits the leukotriene biosynthesis. It is a white to off-white powder that is freely soluble in methanol, ethanol, and water and practically insoluble in acetonitrile.
A montelukast sodium salt is a substance which exhibits efficacy of Singulair (available from Korean MSD) generally used for the treatment of asthma as well as for the symptoms associated with allergic rhinitis, which is pharmaceutically known as a leukotriene receptor antagonist. Leukotrienes produced in vivo by metabolic action of arachidonic acid include LTB4, LTC4, LTD4 and LTE4. Of these, LTC4, LTD4 and LTE4 are cysteinyl leukotrienes (CysLTs), which are clinically essential in that they exhibit pharmaceutical effects such as contraction of airway muscles and smooth muscles and promotion of secretion of bronchial mucus.
Montelukast sodium salt is a white and off-white powder which has physical and chemical properties that it is well soluble in ethanol, methanol and water and is practically insoluble in acetonitrile.
A conventionally known method for preparing a montelukast sodium salt is disclosed in EP Patent No. 480,717. However, the method in accordance with the EP Patent requires processes for introducing and then removing a tetrahydropyranyl (THP) protecting group and purification by chromatography, thus being disadvantageously unsuitable for mass-production. In addition, the method disadvantageously requires investment in high-cost equipment, for example, to obtain amorphous final compounds by lyophilization.
Meanwhile, U.S. Pat. No. 5,614,632 discloses an improved method for preparing a montelukast sodium salt by directly reacting a methanesulfonyl compound (2) with 1-(lithium mercaptomethyl)cyclopropaneacetic acid lithium salt, without using the tetrahydropyranyl protecting group used in EP Patent No. 480,717, purifying in the form of a dicyclohexylamine salt by adding dicyclohexylamine to the reaction solution, and converting the salt into a montelukast sodium salt (1).
However, the method in accordance with the US patent should use n-butyl lithium as a base in the process of preparing the 1-(lithium mercaptomethyl)cyclopropaneacetic acid lithium salt and thus requires an improved process due to drawbacks that n-butyl lithium is dangerous upon handling and is an expensive reagent.
PCT International Patent Laid-open No. WO 2005/105751 discloses a method for preparing a montelukast sodium salt, comprising coupling methyl 1-(mercaptomethyl)cyclopropane acetate (3) used in step 10 shown in Example 146 of EP Patent 480,717 with a methanesulfonyl compound (2) in the presence of a solvent/cosolvent/base, performing hydrolysis, recrystallizing the resultingmontelukast acid (4) in the presence of a variety of solvents to obtain highly puremontelukast acid (4), and converting the same into a montelukast sodium salt (1).
In addition, WO 2005/105751 claims that, in the coupling reaction, one is selected from tetrahydrofurane and dimethylcarbonate as a solvent, a highly polar solvent is selected from dimethylformamide, dimethylacetamide and N-methylpyrrolidone as a cosolvent, and one is selected from sodium hydroxide, lithium hydroxide, sodium hydride, sodium methoxide, potassium tert-butoxide, lithium diisopropylamine and quaternary ammonium salts, as a base.
However, WO 2005/105751 discloses that, since the coupling reaction requires use of a mixed solvent and the mixed solvent is different from the solvent used for hydrolysis, a process for removing the cosolvent through distillation under reduced pressure or extraction is further required prior to hydrolysis.
Further, in accordance with the method of WO 2005/105751, recrystallization is performed in the presence of a variety of solvents in order to obtain a highly puremontelukast acid (4) and the resulting recrystallization yield is varied in a range of 30 to 80%, depending on the solvent. In the case where desired purity is not obtained, recrystallization is repeated until montelukast acid (4) with a desired purity can be obtained. Disadvantageously, the method causes deterioration in overall yield.
European Patent No. 480,717 discloses montelukast sodium and its preparation starting with the hydrolysis of its ester derivative to the crude sodium salt, acidification of the crude to montelukast acid, and purification of the crude acid by column chromatography to give montelukast acid as an oil. The resulting crude oil in ethanol was converted to montelukast sodiumby the treatment with an equal molar aqueous sodium hydroxide solution. After removal of the ethanol, the montelukastsodium was dissolved in water and then freeze dried. The montelukast sodium thus obtained is of a hydrated amorphous form as depicted in FIG. 2.
The reported syntheses of montelukast sodium, as pointed out by the inventor in EP 737,186, are not suitable for large-scale production, and the product yields are low. Furthermore, the final products, as the sodium salts, were obtained as amorphous solid which are often not ideal for pharmaceutical formulation. Therefore, they discloses an efficient synthesis of montelukastsodium by the use of 2-(2-(3-(S)-(3-(7-chloro-2-quinolinyl)ethenyl)phenyl)-3-methanesulfonyloxypropyl)phenyl)-2-propanol to couple with the dilithium salt of 1-(mercaptomethyl)cyclopropaneacetic acid. The montelukast acid thus obtained is converted to the corresponding dicyclohexylamine salt and recrystallized from a mixture of toluene and acetonitrile to obtain crystallinemontelukast sodium. This process provides improved overall product yield, ease of scale-up, and the product sodium salt in crystalline form.
According to the process described in EP 737,186, the chemical as well as optical purities of montelukast sodium depends very much on the reaction conditions for the mesylation of the quinolinyl diol with methanesulfonyl chloride. For instance, the reaction temperature determinates the chemical purity of the resulting coupling product montelukast lithium, due to the fact that an increase in the reaction temperature resulted in decreased selectivity of mesylation toward the secondary alcohol. Mesylation of the tertiary alcohol occurred at higher temperature will produce, especially under acidic condition, the undesired elimination product, the styrene derivative. This styrene impurity is difficult to remove by the purification procedure using DCHA salt formation; while excess base, butyl lithium in this case, present in the reaction mixture causes the formation of a cyclization by-product, which will eventually reduce the product yield.
PCT WO 2005/105751 discloses an alternative process for preparing montelukast sodium by the coupling of the same mesylate as disclosed in ‘186 patent with 1-(mercaptomethyl)cyclopropane alkyl ester in the presence of a base. In this patent, the base butyl lithium, a dangerous and expensive reagent, is replaced with other milder organic or inorganic base. However, the problem concerning the formation of the styrene impurity is still not resolved.
CA 2649189 A1
Process for the manufacture of 1-[[[(1R)-1-[3-[(1E)-2-(7-chloro-2-quinolinyl)ethenyl]phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]thio]methyl]cyclopropane acetic acid, sodium salt [montelukast sodium (I)] consisting of: i. Converting methyl 1-(mercaptomethyl)-cyclopropaneacetate to a metal salt (X) using a metal hydroxide, ii. Subjecting the metal salt (X) to monometallation to provide a dimetallide (XI). iii. Converting a diol of formula (II) to a mesylate of formula (III) and reacting (III) in situ with (XI) affordin the metal salt of 1-[[[(1R)-1-[3-[(1E)-2-(7-chloro-2-quinolinyl)ethenyl]phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]thio]methyl]cyclopropane acetic acid. iv. Reacting the metal salt in-situ with a base and purifying to afford an amine salt (XII). v. Treating (XII) with a sodium base and precipitating out montelukast sodium (I).
more info
European Patent No. 480,717 disclose the montelukast and its preparation method first be hydrolyzed to the crude ester derivatives sodium, then this crude product was acidified to montelukast acid (montelukastacid), Finally, column chromatography purification of this crude acid into oily montelukast acid. This oilMontelukast acid in ethanol, by equimolar amounts of sodium hydroxide solution and converted to montelukast sodium. The ethanol was removed aftermontelukast sodium dissolved in water, followed by freeze-drying. Finally obtainedmontelukast shown in Figure 2 is amorphous hydrated.
The invention, in European Patent No. 737,186 points out, thismontelukast synthesis method is not suitable for mass production, and the low yield. Moreover, the resulting amorphous solid salt, are generally not used in pharmaceutical formulations. Therefore, they disclose the synthesis of an effective method of montelukast sodium, which uses 2 – (2 – (3 – (S) – (3 – (7 – chloro-2 – quinolinyl) ethenyl) phenyl) -3 – methylsulfonyl) phenyl) -2 – propanol and 1 – (methylthio alcohol) cyclopropane coupling the lithium salt of acetic acid, the resulting Montelukast acid is converted into a corresponding bicyclic hexyl amine salt, and from a mixture of toluene and acetonitrile recrystallization to prepare crystalline montelukast. This method greatly improves the productivity, ease of mass production, and the product is crystalline sodium salt.
According to European Patent No. 737,186 described method for preparingmontelukast chemical purity and optical purity depends largely quinoline diol with methanesulfonyl chloride in the reaction between the mesylated condition. For example, the reaction temperature resulted in an increase of the secondary alcohols methanesulfonyl selective reduction, the reaction temperature determines the coupling product (montelukast lithium) chemical purity. Occurs at a higher temperature mesylation tertiary alcohols, in particular under acidic conditions, will produce impurities, such as styrene derivatives. This impurity is difficult styrene generated by using the DCHA salt (DCHA salt formation) in the purification process to remove; present in the reaction mixture and excess base, butyl lithium cyclized by-products resulting in the formation will eventually reduce the yield of the product.
W02005/105751 disclose another preparation method of montelukast sodium, which is the methanesulfonic acid (European Patent No. 737,186 is the same) in an alkaline state where 1_ (methyl mercaptan yl) cyclopropyl alkyl ester and coupling thereof. In this patent, the dangerous and expensive alkaline-butyl lithium reagent, is replaced by other more moderate organic or inorganic base. However, the formation of styrene impurity problem is still not resolved
- Lipkowitz, Myron A. and Navarra, Tova (2001) The Encyclopedia of Allergies (2nd ed.) Facts on File, New York, p. 178, ISBN 0-8160-4404-X
- “Asthma / Allergy “. Mascothealth.com. Retrieved 9 April 2011.
- http://www.merckfrosst.ca/mfcl/en/corporate/research/accomplishments/singulair.html
- “Montelukast Sodium”. The American Society of Health-System Pharmacists. Retrieved 3 April 2011.
- FDA Investigates Merck Drug-Suicide Link
- Updated Information on Leukotriene Inhibitors: Montelukast (marketed as Singulair), Zafirlukast (marketed as Accolate), and Zileuton (marketed as Zyflo and Zyflo CR). Food and Drug Administration. Published June 12, 2009. Accessed June 13, 2009.
- Rubenstein, Sarah (April 28, 2008). “FDA Sneezes at Claritin-Singulair Combo Pill”. The Wall Street Journal.
- Schering-Plough press release – Schering-Plough/MERCK Pharmaceuticals Receives Not-Approvable Letter from FDA for Loratadine/Montelukast
- 5,565,473
- Singular patent details
- “FDA approves first generic versions of Singulair to treat asthma, allergies”. 03 August 2012. Retrieved 15 August 2012.
- “U.S. Reexamines Merck’s Singulair Patent”. Thompson Reuters. May 28, 2009.
- “Merck Says U.S. Agency Upholds Singulair Patent”. Thompson Reuters. December 17, 2009.
GENERAL METHOD1

………………………………..
READ ABOUT S ISOMER
http://file.scirp.org/Html/8-2200440_26971.htm
…………………….
Improved Process for the Preparation of Montelukast: Development of an Efficient Synthesis, Identification of Critical Impurities and Degradants


Codexis, Inc., Redwood City, USA and Arch PharmaLabs Limited, Mumbai, India
Development of a Biocatalytic Process as an Alternative to the (-)-DIP-Cl-Mediated Asymmetric Reduction of a Key Intermediate of Montelukast
Org. Process Res. Dev. 2010, 14: 193-198

Montelukast sodium (Singulair®) is a leukotriene receptor antagonist prescribed for the treatment of asthma and allergies. Workers at Codexis used directed evolution and high-throughput screening to engineer a robust and efficient ketoreductase enzyme (CDX-026) that accomplished the asymmetric reduction of ketone A, which is essentially water insoluble, at a loading of 100 g/L in the presence of ca. 70% organic solvents at 45 ˚C. The (S)-alcohol B was obtained in >95% yield in >99.9% ee and in >98.5% purity on a >500 mol scale.
The enzymatic reduction entails the reversible transfer of a hydride from isopropanol to the ketone A with concomitant formation of acetone. The reaction is driven to completion by the fortuitous crystallization of the monohydrate B. The four-step conversion of B into montelukast sodium is described in the Merck process patent (M. Bhupathy, D. R. Sidler, J. M. McNamara, R. P. Volante, J. J. Bergan US 6320052, 2001). This biocatalytic reduction is superior to the reduction of A with (-)-DIPCl previously used in the manufacture of montelukast
















