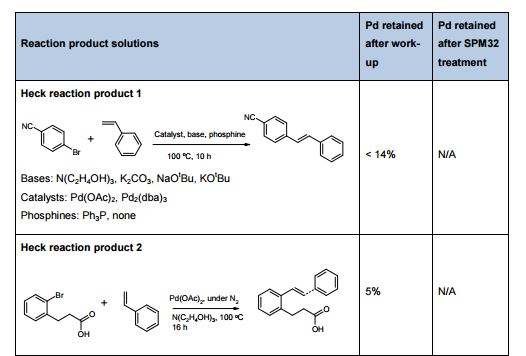
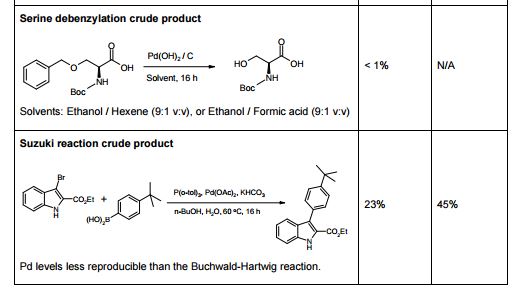
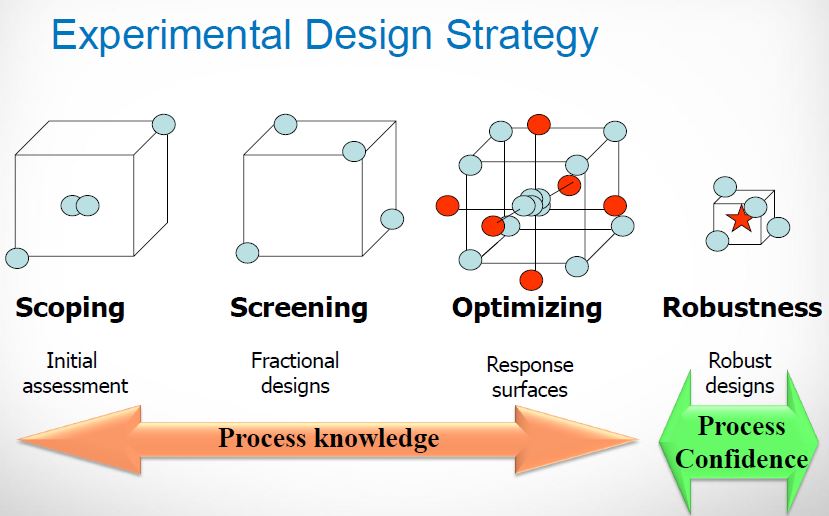
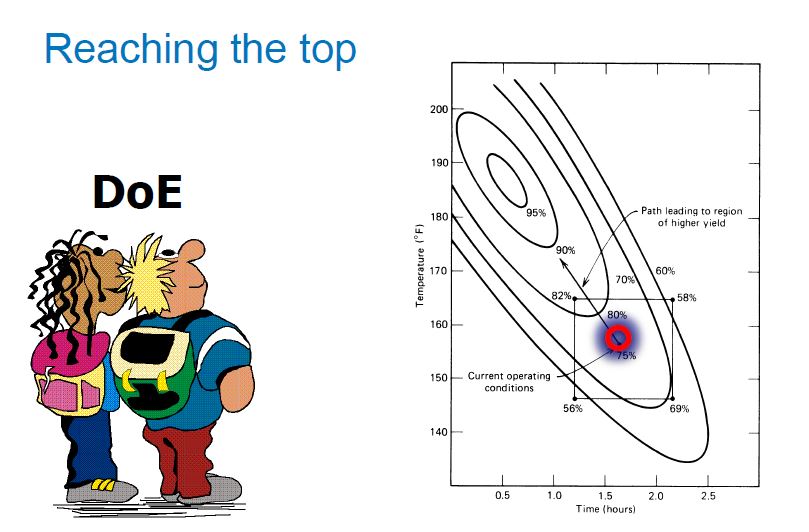
The influence of four parameters (temperature, scavenging time, amount of scavenger, and concentration of palladium in the solution) on the efficiency of Pd removal from a cross-coupling reaction, using a commercially available Pd scavenger, SPM32, was studied. The DoE-based method employed yielded more information than is readily attainable from standard adsorption isotherms and kinetics experiments. The optimal regime of scavenging was identified; intuitive and nonintuitive effects of temperature, scavenging time, and scavenger amounts were highlighted; and a mathematical model quantifying predicted Pd removal from the synthetic intermediate was built.
link http://pubs.acs.org/doi/abs/10.1021/op5000336
Statistical DoE Approach to the Removal of Palladium from Active Pharmaceutical Ingredients (APIs) by Functionalized Silica Adsorbents
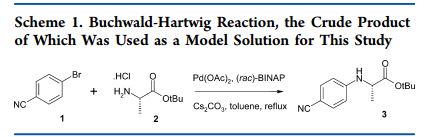
Preparation of tert-butyl 2-[(4-cyanophenyl)amino]propanoate (3).
4-Bromobenzonitrile (18.20 g, 100 mmol), L-alanine tert-butyl ester hydrochloride (21.73 g, 120 mmol), ±BINAP (1.25 g, 2 mmol) and cesium carbonate (48.87 g, 150 mmol) were added to a 3-neck round bottom flask containing a magnetic stirrer. Toluene (167 mL) was added and a reflux condenser, thermometer and a rubber septum were attached. Argon gas was bubbled through as the heterogeneous mixture was warmed to reflux temperature with slow agitation from the magnetic stirrer. Palladium acetate (0.45 g, 2 mmol) was added quickly through one of the side-arm joints and de-gassing was continued for 5 min. The reaction mixture was kept under argon at reflux and the disappearance of 4-bromobenzonitrile was monitored by GC-MS. After 16-24 h, the reaction mixture was filtered through a sinter funnel, washed with toluene and then filtered through a nylon membrane (Sigma Aldrich catalogue no. Z290793, 0.45 µm pore size) and washed with toluene. This crude reaction mixture was used in the DoE matrix without further purification.
Experimental results
NMR δC (62.9 MHz, CDCl3): 172.7, 150.0, 133.7, 120.4, 112.7, 99.3, 82.3, 51.7, 28.0, 18.5 ppm.
NMR δH (250 MHz, CDCl3): 7.39 (2H, d, J = 8.8 Hz, Ph), 6.52 (2H, d, J = 8.8 Hz, Ph), 4.85 (1H, d, J = 7.4 Hz, NH), 4.01 (1H, quintet, J = 7.4 Hz, CH(Me), 1.43 (9H, s, tBu), 1.42 (3H, d, J = 7.4 Hz, Me).
GC/MS: GC method used: hold at 50 °C for 4 min; increase temperature from 50 to 280 °C at 30 °C / min; hold at 280 °C for 5 min. Peaks were recorded and identified as follows: 4-Bromobenzonitrile: 10.05 min. Molecular peak observed at m/z = 181 for the 79Br isotope, m/z = 183 for the 81Br isotope L-alanine tert-butyl ester hydrochloride: not observed. Retention time less than 5 min, peak lost within the solvent front. Product: 13.64 min. Molecular peak observed at m/z = 246, main fragment at m/z = 146 (M – CO2 t Bu) Side product (not identified or quantified, minor peak): 14.56 min.

Jan Recho
Systems developer at Clearsy
Corresponding Author *E-mail: jan.recho@phosphonics.com.
Experience
Systems Developer
Clearsy
– Present (1 year 6 months)
Scientist
PhosphonicS
– (3 years 6 months)
• Design and synthesis of silica supported transition metals scavengers and catalysts (Pd, Rh, Ru) for the pharmaceutical industry.
• Management of fine chemistry customer projects
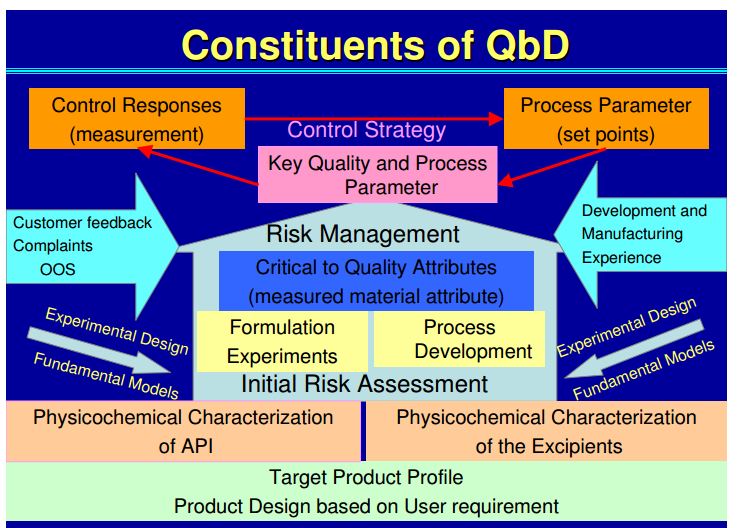
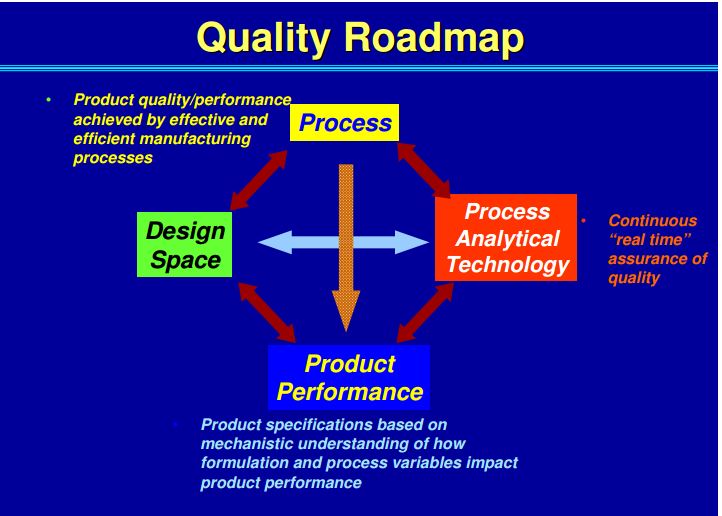
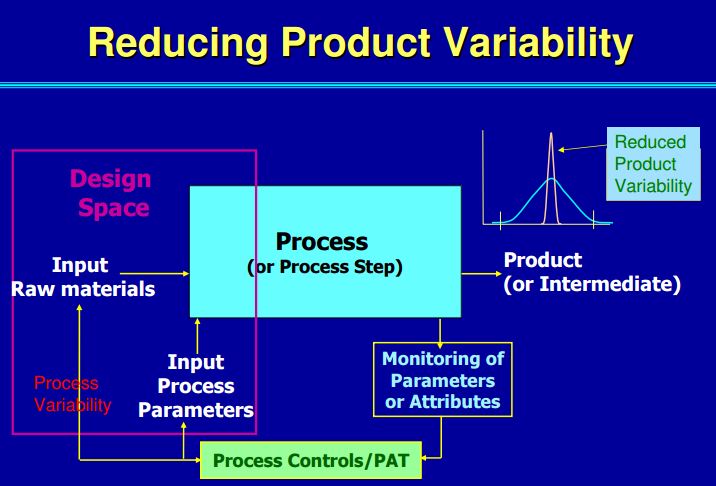
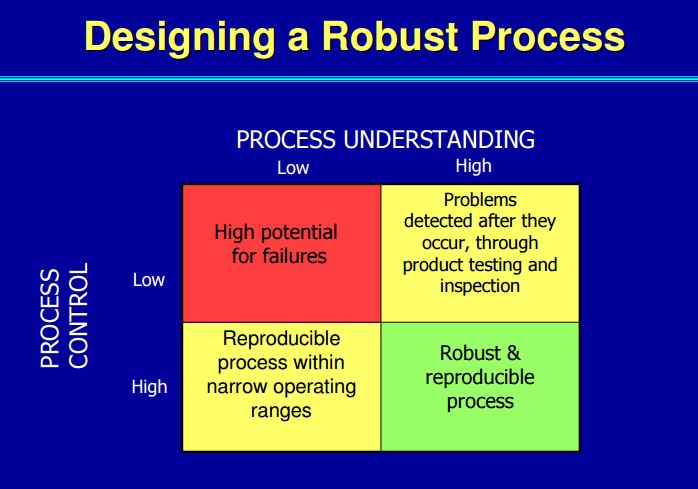
• Process optimisation (Quality by Design – QbD, Design of Experiments – DoE).
• Business development activities for the French market
Junior researcher
Institut des Matériaux de Nantes
– (4 years 8 months)Nantes Area, France
Synthesis and characterisation of a cellulose derived, organosilane-based, bio-material for cartilage growth.
Work in imidazolium and pyridinium ionic liquids.