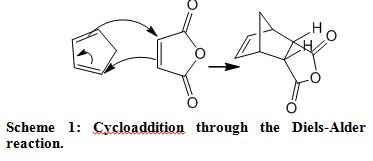
ENDO EXO STORY…….cis-norborene-5,6-endo-dicarboxylic anhydride

You will react cyclopentadiene with maleic anhydride to form the Diels-Alder product below. This Diels-Alder reaction produces almost solely the endo isomer upon reaction at ambient temperature.

The preference for endo–stereochemistry is “observed” in most Diels-Alder reactions. The fact that the more hindered endo product is formed puzzled scientists until Woodward, Hoffmann, and Fukui used molecular orbital theory to explain that overlap of the p orbitals on the substituents on the dienophile with p orbitals on the diene is favorable, helping to bring the two molecules together.
Hoffmann and Fukui shared the 1981 Nobel Prize in chemistry for their molecular orbital explanation of this and other organic reactions. In the illustration below, notice the favorable overlap (matching light or dark lobes) of the diene and the substituent on the dienophile in the formation of the endo product:

Oftentimes, even though the endo product is formed initially, an exo isomer will be isolated from a Diels-Alder reaction. This occurs because the exo isomer, having less steric strain than the Endo , is more stable, and because the Diels-Alder reaction is often reversible under the reaction conditions. In a reversible reaction, the product is formed, reverts to starting material, and forms again many times before being isolated.
The more stable the product, the less likely it will be to revert to the starting material. The isolation of an exo product from a Diels-Alder reaction is an example of an important concept: thermodynamic vs kinetic control of product composition. The first formed product in a reaction is called the kinetic product. If the reaction is not reversible under the conditions used, the kinetic product will be isolated. However, if the first formed product is not the most stable product and the reaction is reversible under the conditions used, then the most stable product, called the thermodynamic product, will often be isolated.
The NMR spectrum of cis-5-norbornene-2,3-endo-dicarboxylic anhydride is given below:

Cis-Norbornene-5,6-endo-dicarboxylic anhydride
Cyclopentadiene was previously prepared through the cracking of dicyclopentadiene and kept under cold conditions. In a 25 mL Erlenmeyer flask, maleic anhydride (1.02 g, 10.4 mmol) and ethyl acetate (4.0 mL) were combined, swirled, and slightly heated until completely dissolved. To the mixture, ligroin (4 mL) was added and mixed thoroughly until dissolved. Finally, cyclopentadiene (1 mL, 11.9 mmol) was added to the mixture and mixed extensively. The reaction was cooled to room temperature and placed into an ice bath until crystallized. The crystals were isolated through filtration in a Hirsch funnel. The product had the following properties: 0.47 g (27.6% yield) mp: 163-164 °C (lit: 164 °C). 1H NMR (CDCl3, 300 MHz) δ: 6.30 (dd, J=1.8 Hz, 2H), 3.57 (dd, J=7.0 Hz, 2H), 3.45 (m, 2H), 1.78 (dt, J=9.0,1.8 Hz, 1H), 1.59 (m, 1H) ppm. 13C NMR (CDCl3, 75Hz) δ: 171.3, 135.5, 52.7, 47.1, 46.1 ppm. IR 2982 (m), 1840 (s), 1767 (s), 1089 (m) cm-1.
When combining the reagents, a cloudy mixture was produced and problems arose in the attempt to completely dissolve the mixture. After heating for about 10 minutes and magnetically stirring, tiny solids still remained. The undissolved solids were removed form the hot solution by filtration and once they cooled, white crystals began to form. Regarding the specific reaction between cyclopentadiene and maleic anhydride, the endo isomer, the kinetic product, was formed because the experiment was directed under mild conditions. The exo isomer is the thermodynamic product because it is more stable.3
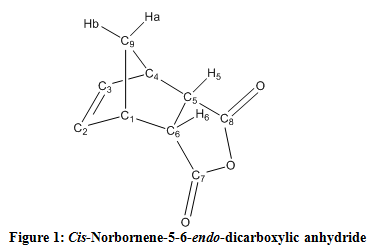
The 1H NMR spectum exhibited a doublet of doublets at 3.57 ppm for the endo isomer. The exo isomer would possess a triplet around 3.50 ppm due to the difference in dihedral angle between the hydrogen molecules of H-1 and H-4, and H-5 and H-6 (Figure 1). A peak at 3.00 ppm would appear in the exo isomer spectra as opposed to a peak at 3.60 ppm as shown in the observed endo product.3 This is because of the interaction and coupling with the H-5 and H-6, as displayed in Figure 1.
Through the Diels-Alder reaction, 27.6% yield of cis-Norbornene-5,6-endo-dicarboxylic anhydride was produced. The distinction of the presence of the endo isomer was proven by analyzing physical properties of both possible isomers.
link
http://orgspectroscopyint.blogspot.in/2014/08/cis-norborene-56-endo-dicarboxylic.html
ORGANIC SPECTROSCOPY INTERNATIONAL


DR ANTHONY MELVIN CRASTO Ph.D
web link


 amcrasto@gmail.com
amcrasto@gmail.com










