DDD 107498, DDD 498
PATENT WO 2013153357, US2015045354
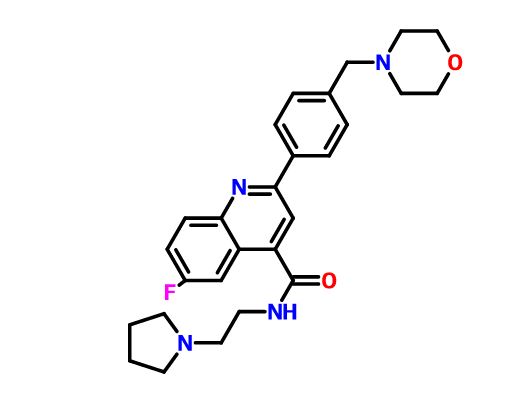
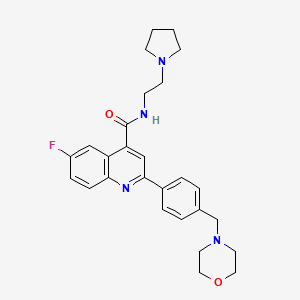
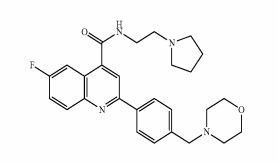
6-Fluoro-2-[4-(morpholinomethyl)phenyl]-N-(2-pyrrolidin-1-ylethyl)quinoline-4-carboxamide
6-Fluoro-2-[4-(4-morpholinylmethyl)phenyl]-N-[2-(1-pyrrolidinyl)ethyl]-4-quinolinecarboxamide
4-Quinolinecarboxamide, 6-fluoro-2-[4-(4-morpholinylmethyl)phenyl]-N-[2-(1-pyrrolidinyl)ethyl]-
CAS 1469439-69-7
CAS 1469439-71-1 SUCCINATE
| MF C27H31FN4O2 | |
| MW | 462.559043 g/mol |
|---|
- 6-fluoro-2-[4-(morpholin-4-ylmethyl)phenyl]-N-(2-pyrrolidin-1-ylethyl)quinoline-4-carboxamide
- Originator Medicines for Malaria Venture; University of Dundee
- Class Small molecules
- Mechanism of Action Protein synthesis inhibitors
Highest Development Phases
- No development reported Malaria
Most Recent Events
- 16 Jul 2016 No recent reports of development identified for preclinical development in Malaria in United Kingdom
- 01 Apr 2015 DDD 498 licensed to Merck Serono worldwide for the treatment of Malaria
| Inventors | Ian Hugh Gilbert, Neil Norcross, Beatriz Baragana Ruibal, Achim Porzelle |
| Original Assignee | University Of Dundee |
Prof Ian Gilbert:
Head of Biological Chemistry and Drug Discovery
BCDD, College of Life Sciences, University of Dundee, DD1 5EH, UK
Tel: +44 (0) 1382-386240


Merck Serono and MMV sign agreement to develop potential antimalarial therapy
Agreement further diversifies MMV’s partner base, strengthening our antimalarial research and development portfolio
 Photo © Merck Serono
Photo © Merck Serono
Merck Serono, the biopharmaceutical business of Merck, and MMV announced today that an agreement has been signed for Merck Serono to obtain the rights to the investigational antimalarial compound DDD107498 from MMV. This agreement underscores the commitment of Merck Serono to provide antimalarials for the most vulnerable populations in need.
“This agreement strengthens our Global Health research program and our ongoing collaboration with Medicines for Malaria Venture,” said Luciano Rossetti, Executive Vice President, Global Head of Research & Development at Merck Serono. “MMV is known worldwide for its major contribution to delivering innovative antimalarial treatments to the most vulnerable populations suffering from this disease, and at Merck Serono we share this goal.”
DDD107498 originated from a collaboration between MMV and the University of Dundee Drug Discovery Unit, led by Prof. Ian Gilbert and Dr. Kevin Read. The objective of the clinical program is to demonstrate whether the investigational compound exerts activity on a number of malaria parasite lifecycle stages, and remains active in the body long enough to offer potential as a single-dose treatment against the most severe strains of malaria.
While development and commercialization of the compound is under Merck Serono’s responsibility, MMV will provide expertise in the field of malaria drug development, including its clinical and delivery expertise, and provide access to its public and private sector networks in malaria-endemic countries.
Merck Serono has a dedicated Global Health R&D group working to address key unmet medical needs related to neglected diseases, such as schistosomiasis and malaria, with a focus on pediatric populations in developing countries. Its approach is based on public-private partnerships and collaborations with leading global health institutions and organizations in both developed and developing countries.
“Working with partners like Merck Serono is critical to the progress of potential antimalarial compounds, like DDD107498, through the malaria drug pipeline,” said Dr. Timothy Wells, Chief Scientific Officer at MMV. “Their Global Health Program is gaining momentum and we need more compounds to tackle malaria, a disease that places a huge burden on the world’s most vulnerable populations. DDD107498 holds great promise and we look forward to working with the Merck Serono team through the development phase.”
According to the World Health Organization, there were an estimated 198 million cases of malaria worldwide in 2013, and an estimated 584,000 deaths, primarily in young children from the developing world. The launch of the not-for-profit research foundation, MMV, in 1999 and a number of collaborations and partnerships, including those with Merck Serono, has contributed to reducing the major gap in malaria R&D investment and subsequent dearth of new medicines.
“It’s hugely encouraging to see the German pharmaceutical industry increasing their engagement in the development of novel antimalarials,” said global malaria expert Prof. Dr. Peter Kremsner, Director of the Institute for Tropical Medicine at the University of Tübingen, Germany. “The Merck Serono and MMV collaboration to develop DDD107498 is a great step. It’s a compound that offers lots of promise so I’m excited to see how it progresses.
Scots scientists in ‘single dose’ malaria treatment breakthrough
An antimalarial drug that could treat patients was discovered by Dundee university scientists
Scientists have discovered an antimalarial compound that could treat malaria patients in a single dose and help prevent the spread of the disease from infected people.
The compound DDD107498 also has the potential to treat patients with malaria parasites resistant to current medications, researchers say.
Scientists hope it could lead to treatments and protection against the disease, which claimed almost 600,000 lives amid 200 million reported cases in 2013.
The compound was identified through a collaboration between the University of Dundee’s drug discovery unit (DDU) and the Medicines for Malaria Venture (MMV), a separate organisation.
The compound is now undergoing further safety testing with a view to entering human clinical trials within the next year.
Details of the discovery have been published in the journal Nature.
Professor Ian Gilbert, head of chemistry at the DDU, who led the team that discovered the compound, said: “The publication describes the discovery and profiling of this exciting new compound.
“It reveals that DDD107498 has the potential to treat malaria with a single dose, prevent the spread of malaria from infected people and protect a person from developing the disease in the first place.
“There is still some way to go before the compound can be given to patients. However, we are very excited by the progress that we have made.”
The World Health Organisation reports that there were 200 million clinical cases of malaria in 2013, with 584,000 people dying from the disease. Most of these deaths were children under the age of five and pregnant women.
MMV chief executive officer Dr David Reddy said: “Malaria continues to threaten almost half of the world’s population – the half that can least afford it.
“DDD107498 is an exciting compound since it holds the promise to not only treat but also protect these vulnerable populations.
“The collaboration to identify and progress the compound, led by the drug discovery unit at the University of Dundee, drew on MMV’s network of scientists from Melbourne to San Diego.”The publication of the research is an important step and a clear testament to the power of partnership.”
MMV selected DDD107498 to enter preclinical development in October 2013 following the recommendation of its expert scientific advisory committee.
Since then, with MMV’s leadership, large quantities of the compound have been produced and it is undergoing further safety testing with a view to entering human clinical trials within the next year.
Merck Serono has recently obtained the right to develop and, if successful, commercialise the compound, with the input of MMV’s expertise in the field of malaria drug development and access and delivery in malaria-endemic countries.
Dr Michael Chew from the Wellcome Trust, which provides funding for the DDU and MMV, said: “The need for new antimalarial drugs is more urgent than ever before, with emerging strains of the parasite now showing resistance against the best available drugs.
“These strains are already present at the Myanmar-Indian border and it’s a race against time to stop resistance spreading to the most vulnerable populations in Africa.
“The discovery of this new antimalarial agent, which has shown remarkable potency against multiple stages of the malaria lifecycle, is an exciting prospect in the hunt for viable new treatments.”
PAPER


Discovery of a Quinoline-4-carboxamide Derivative with a Novel Mechanism of Action, Multistage Antimalarial Activity, and Potent in Vivo Efficacy

A single-dose treatment against malaria worked in mice to cure them of the disease. The drug also worked to block infection in healthy mice and to stop transmission, according to a study published in Nature today. The fact that the drug can act against so many stages of malaria is pretty new, but what’s even more exciting is the compound’s mode of action: it kills malaria in a completely new way, researchers say. The feature would make it a welcome addition to our roster of antimalarials — a roster that’s threatened by drug resistance.
RESEARCHERS SIFTED THROUGH A LIBRARY OF ABOUT 4,700 COMPOUNDS TO FIND THIS ONE
Malaria is an infectious disease that’s transmitted through mosquito bites; it’s also a leading cause of death in a number of developing countries. Approximately 3.4 billion people live in areas where malaria poses a real threat. As a result, there were 207 million cases of malaria in 2012 — and 627,000 deaths. There are drugs that can be used to prevent malaria, and even treat it, but drug resistance is halting the use of certain treatments in some areas.
A long search
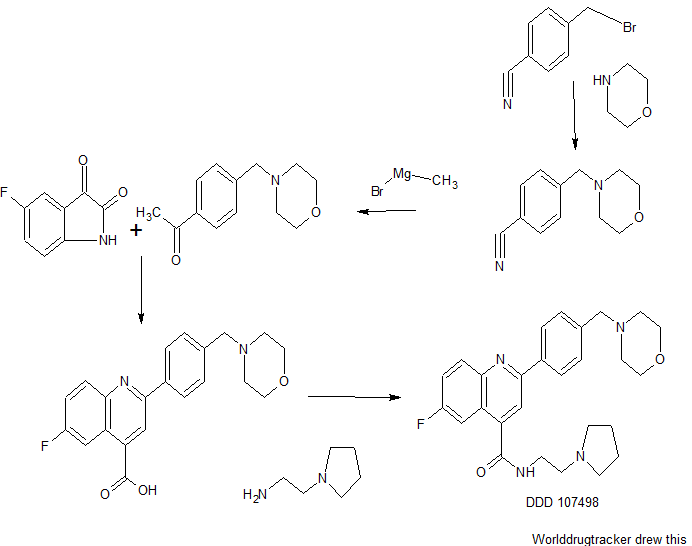
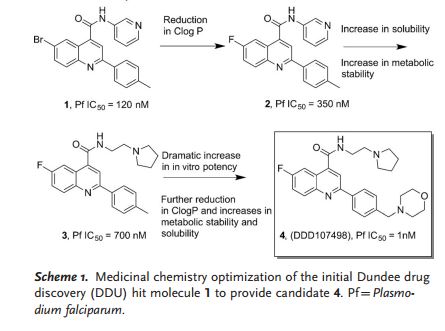
Searching for a new drug is all about trial and error. To find this particular compound, researchers sifted through a library of about 4,700 compounds, testing them to see if they were capable of killing the malaria parasite in a lab setting. When they found something that worked, they tweaked the drug candidate to see if it could perform more effectively. “We went through a lot of these cycles of testing and designing new compounds,” says Ian Gilbert, a medicinal chemist at the University of Dundee in the UK, and a co-author of the study. “Eventually we optimized to the compound which is the subject of the paper.” For now, that compound’s unwieldy name is DDD107498.
To make sure DDD107498 really had potential, the researchers tested it on mice that had already been infected with malaria. A single dose was enough to provoke a 90 percent reduction in the number of parasites in their blood. The scientists also gave the compound to healthy mice that were subsequently exposed to malaria. DDD107498 helped the mice evade infection with a single dose, but it’s unclear how long that effect would last in humans. Finally, the researchers looked at whether the compound could prevent the transmission from an infected mouse to a mosquito. A day after receiving the treatment, mice were put in contact with mosquitoes. The scientists noted a 91 percent reduction in infected mosquitoes.
“IT HAS THE ABILITY TO BE A ONE-DOSE [DRUG], IN COMBINATION WITH ANOTHER MOLECULE.”
“What’s exciting about this molecule is obviously the fact that it has the ability to be a one-dose [drug], in combination with another molecule to cure blood stage malaria,” says Kevin Read, a drug researcher also at the University of Dundee and a co-author of the study. The fact that the compound has the ability to block transmission and protect against infection is equally thrilling. But the way in which DDD107498 kills malaria might be its most interesting feature. It halts the production of proteins — which are necessary for the parasite’s survival. No other malaria drug does that right now, Read says. “So, in principle, there’s no resistance out there already to this mechanism.”
The drug hasn’t been tested in humans yet, so it may not be nearly as good in the field. But Read says DDD107498 looks promising. “From all the pre-clinical or non-clinical data we’ve generated, it is comparable or better than any of the current marketed anti-malarials in those studies.” And at $1 per treatment, the price of the drug should fall “within the range of what’s acceptable,” he says.
“It looks like an excellent study, and the results look very important,” says Philip Rosenthal, a malaria drug researcher at The University of California-San Francisco who didn’t participate in the study. This is a big shift for Rosenthal’s field. Five years ago, “we had very little going on in anti-malarial drug discovery,” he says. Now, there’s quite a bit going on for malaria researchers, and a number of promising compounds are moving along. DDD107498 “is another player, and it’s got a number of positive features,” he says.
OTHER TREATMENTS HAVE TO BE TAKEN FOR A FEW DAYS
One of the features is the drug’s potency. It’s very active against cultured malaria parasites, Rosenthal says. But what’s perhaps most intriguing about DDD107498 is that the drug works against the mechanism that enables protein synthesis the malaria parasite’s cells. No other malaria drug does that right now, Read says. “Considering challenges of treating malaria, which is often in rural areas and developing countries, a single dose would be a big plus,” he says. “In addition, because of it’s long half life, it may also work to prevent malaria with once a week dosing, which is also a benefit.”
Still, no drug is perfect. The data suggests that DDD107498 doesn’t kill malaria as quickly as some other drugs, Rosenthal says. And when the researchers tested it to see how long it might take for resistance to develop, the results weren’t as promising as he would like. The parasites figured out a way to become resistant to the compound “relatively easily,” he says. That shouldn’t be “deal-killer,” however. “Its slow onset of action probably means it should be combined with a faster-acting drug,” he says.
BUT IT’S SLOW-ACTING
The compound is going through safety testing now. If everything goes well, it should hit human trials within the next year, Read says. Chances are, it will have to be used in combination with other malaria drugs, Gilbert says. “All anti-malarials are given in combination because it slows down resistance.”
“When you’re treating infectious diseases, you know that drug resistance is always a potential problem, so having a number of choices to treat malaria is a good thing,” Rosenthal says. In this case, the drug’s new mode of action may hold lead to an entirely new weapon against malaria. “Obviously it’s got a long way to go,” Read says. But the compound is “very exciting,” nonetheless.
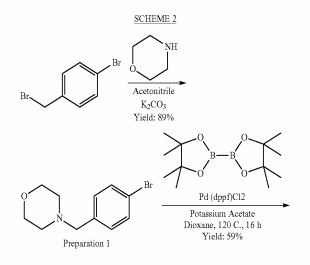
SCHEME 1
SCHEME 4 B
Yield: 72% Yield: 70% Preparation 6
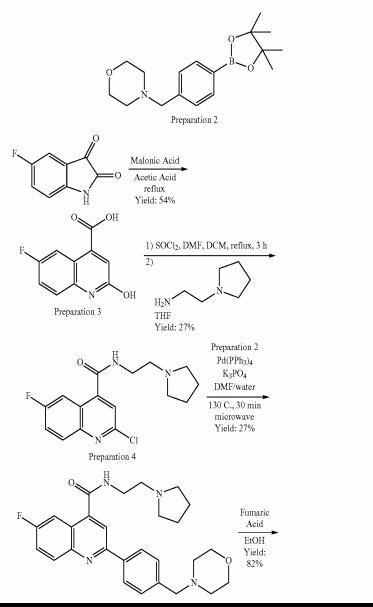
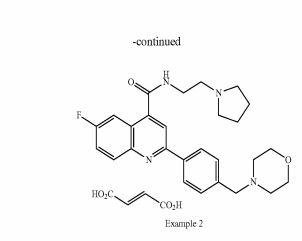
Example 1 : 6-Fluoro-2-r4-(morpholinomethyl)phenyll-N-(2-pyrrolidin-1-ylethyl)quinoline- 4-carboxamide, Example compound 1 in Scheme 2
In a sealed microwave tube, a suspension of 2-chloro-6-fluoro-N-(2-pyrrolidin-1- ylethyl)quinoline-4-carboxamide (preparation 4) (2.00 g, 6 mmol), [4- (morpholinomethyl)phenyl]boronic acid, hydrochloride, available from UORSY, (3.20 g, 12 mmol), potassium phosphate (2.63 g, 12 mmol) and tetrakis(triphenylphosphine)palladium (0) (0.21 g, 0.19 mmol) in DMF/Water 3/1 (40 ml) was heated at 130°C under microwave irradiation for 30 min. The reaction was filtered through Celite™ and solvents were removed under reduced pressure. The resulting residue was taken up in DCM (150 ml) and washed twice with NaHC03 saturated aqueous solution (2 x 100 ml). The organic layer was separated, dried over MgS04and concentrate to dryness under reduced pressure. The reaction crude was purified by flash column chromatography using an 80 g silica gel cartridge and eluting with DCM (Solvent A) and MeOH (Solvent B) and the following gradient: 1 min hold 100% A, followed by a 30 min ramp to 10 % B, and then 15 min hold at 10% B. The fractions containing product were pooled together and concentrated to dryness under vacuum to obtain the desired product as an off-white solid (1 g). The product was dissolved in methanol (100 ml) and 3-mercaptopropyl ethyl sulfide Silica (Phosphonics, SPM-32, 60- 200 uM) was added. The suspension was stirred at room temperature over for 2 days and then at 50°C for 1 h. After cooling to room temperature, the scavenger was filtered off and washed with methanol (30 ml). The solvent was removed under reduced pressure and the product was further purified by preparative HPLC. The fractions containing product were pooled together and freeze dried to obtain the desired product as a white solid (0.6 g, 1.3 mmol, Yield 20%).
1 H NMR (500 MHz; CDCI3) δ 1.81-1.84 (m, 4H), 2.50-2.52 (m, 4H), 2.63 (brs, 4H), 2.82 (t, 2H, J = 5.9 Hz), 3.61 (s, 2H), 3.71 (dd, 2H, J = 5.4 Hz, J = 1 1.4 Hz), 3.74-3.76 (m, 4H), 6.84 (brs, 1 H), 7.52-7.57 (m, 3H), 7.97-8.00 (m, 2H), 8.13 (d, 2H, J = 8.2 Hz), 8.21 (dd, 1 H, J = 5.5 Hz, J = 9.2 Hz) ppm . 19 F NMR (407.5 MHz; CDCI3) δ -11 1.47 ppm. Purity by LCMS (UV Chromatogram, 190-450nm) 99 %, rt = 5.7 min, m/z 463 (M+H)+ HRMS (ES+) found 463.2501 [M+H]+, C27H32F1 N402 requires 463.2504.
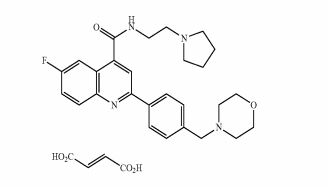
Example 2: 6-Fluoro-2-[4-(morpholinomethyl)phenyl1-N-(2-pyrrolidin-1-ylethyl)quinoline- 4-carboxamide; fumaric acid salt, compound (IB) in Scheme 2
The starting free base (example 1) (0.58 g, 1 mmol) was dissolved in dry ethanol (10 ml) and added dropwise to a stirred solution of fumaric acid (0.15 g, 1 mmol) in dry ethanol (9 ml). The mixture was stirred at room temperature for 1 h. The white precipitate was filtered, washed with ethanol (20 ml) and then dissolved in 10 ml of water and freeze dried to obtain the desired salt as a white solid (0.601 g, 1 mmol, Yield 82%).
1 H NMR (500 MHz; d6-DMSO) δ 1.83-1.86 (m, 4H), 2.41 (brs, 4H), 2.94 (brs, 4H), 3.03 (t, 2H, J = 6.2 Hz), 3.57 (s, 2H), 3.60-3.65 (m, 6H), 6.47 (s, 2H), 7.51 (d, 2H, J = 8.25), 7.74-7.78 (m, 1 H), 8.06 (dd, 1 H, J = 2.9 Hz, J = 10.4 Hz), 8.17 (dd, 1 H, J = 5.7 Hz, J = 9.3 Hz), 8.24-8.26 (m, 3H), 9.24 (t, 1 H, J = 5.5 Hz) ppm. 19 F NMR (407.5 MHz; d6- DMSO) δ -112.30 ppm.
Purity by LCMS (UV Chromatogram, 190-450nm) 99 %, rt = 5.3 min, m/z 463 (M+H)+
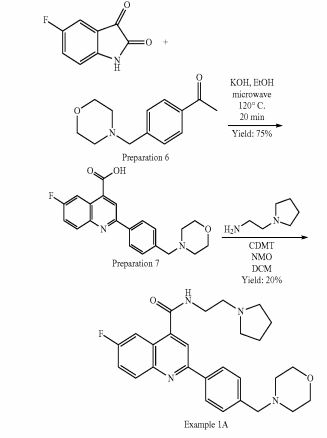
Example 1A: Alternative synthesis of 6-fluoro-2-[4-(morpholinomethyl)phenyl1-N-(2- pyrrolidin-1-ylethyl)quinoline-4-carboxamide, Example compound 1A in Scheme 4
To a stirred suspension of 6-fluoro-2-[4-(morpholinomethyl)phenyl]quinoline-4-carboxylic acid (preparation 7) (2.20 g, 6 mmol) in DCM (100 ml) at room temperature, 2-chloro- 4,6-dimethoxy-1 ,3,5-triazine (CDMT) (1.26 g, 7 mmol) and 4-methylmorpholine (NMO) (1.33 ml, 12 mmol) were added. The reaction mixture was stirred at room temperature for 1 h and then 2-pyrrolidin-1-ylethanamine (0.77 ml, 6 mmol) was added and stirred at room temperature for further 3 h. The reaction mixture was washed with NaHC03 saturated aqueous solution (2x 100 ml) and the organic phase was separated, dried over MgS04 and concentrated under reduced pressure. The resulting residue was absorbed on silica gel and purified by flash column chromatography using an 80 g silica gel cartridge and eluting with DCM (Solvent A) and MeOH (Solvent B) and the following gradient: 2 min hold 100% A followed by a 30 min ramp to 10 %B and then 15 min hold at 10%B. The desired fractions were concentrated to dryness under vacuum to obtain the crude product as a yellow solid (95% purity by LCMS). The sample was further purified by a second column chromatography using a 40 g silica gel cartridge, eluting with DCM (Solvent A) and 10% NH3-MeOH in DCM (Solvent B) and the following gradient: 2 min hold 100% A, followed by a 10 min ramp to 23 % B and then 15 min hold at 23% B. The desired fractions were concentrated to dryness under vacuum to obtain product as a white solid (1 g). Re-crystallisation form acetonitrile (18 ml) yielded the title compound as a white solid (625 mg, 1.24 mmol, 20%).
1 H NMR (500 MHz; CDCI3) δ 1.81-1.84 (m, 4H), 2.50-2.52 (m, 4H), 2.63 (brs, 4H), 2.82 (t, 2H, J = 5.9 Hz), 3.61 (s, 2H), 3.71 (dd, 2H, J = 5.4 Hz, J = 1 1.4 Hz), 3.74-3.76 (m, 4H), 6.84 (brs, 1 H), 7.52-7.57 (m, 3H), 7.97-8.00 (m, 2H), 8.13 (d, 2H, J = 8.2 Hz), 8.21 (dd, 1 H, J = 5.5 Hz, J = 9.2 Hz) ppm .
1 H NMR (500 MHz; d6-DMSO) δ 1.72-1.75 (m, 4H), 2.41 (brs, 4H), 2.56 (brs, 4H), 2.67 (t, 2H, J = 6.6 Hz), 3.49-3.52 (m, 2H), 3.56 (s, 2H), 3.60-3.61 (m, 4H), 7.52 (d, 2H, J = 8.3 Hz), 7.73-7.77 (m, 1 H), 8.07 (dd, 1 H, J = 2.9 Hz, J = 10.4 Hz), 8.18-8.21 (m, 2H), 8.26 (d, 2H , J = 8.3 Hz), 8.85 (t, 1 H, J = 6.6 Hz) ppm.
13C NMR (125 MHz; d6-DMS03) 5 23.2, 38.4, 53.2, 53.5, 54.5, 62.1 , 66.2, 109.0, 109.1 , 1 17.3, 120.1 , 120.3, 124.1 , 124.2, 127.1 , 129.4, 132.2, 132.3, 136.8, 139.9, 142.8, 145.2, 155.3, 159.0, 161 .0, 166.1 ppm.
19 F NM R (500 MHz; d6-DMSO) δ -1 12.47 ppm.
Purity by LCMS (UV Chromatogram, 190-450nm) 99 %, rt = 5.0 min, m/z 463 (M+H)+
Angewandte Chemie, International Edition (2015), 54, (46), 13504-13506

Putting a stop to malaria: Phenotypic screening against malaria parasites, hit identification, and efficient lead optimization have delivered the preclinical candidate antimalarial DDD107498. This molecule is distinctive in that it has potential for use as a single-dose cure for malaria and shows a unique broad spectrum of activity against the liver, blood, and mosquito stages of the parasite life cycle.
Professor Ian Gilbert FRSC
Medicinal Chemistry
I am a medicinal chemist and my research interests are in the design and synthesis of potential drugs. The mainstay of my work is synthetic medicinal chemistry as part of the Drug Discovery Unit (DDU). Where possible we make extensive use of molecular modeling to guide our synthetic efforts. I have a particular interests in the following aspects of drug discovery:
Neglected diseases such as human African trypanosomiasis, leishmaniasis and malaria.
Chemical validation of drug targets, including novel targets for which there is little or no precedence for drug discovery.
Novel approaches to and paradigms for drug discovery.
Mode of action studies and target identification.
I am Head of Chemistry in the DDU. Our main focuses are on neglected diseases and novel drug targets. The neglected diseases we are tackling are malaria, tuberculosis and the kinetoplastid diseases. We use both target-based approaches and phenotypic approaches (whole parasite screening). We have had particular success in validating the enzyme N-myristoyltransferase as a drug target in human African trypanosomiasis, and in identifying and optimising phenotypic hits. In our novel targets area, we aim to validate novel areas of biology as potential drug targets.
Dr Neil Norcross


Achim Porzelle
///////////DDD107498, DDD 107498, PRECLINICAL, DUNDEE, MALARIA, DDD 498, Achim Porzelle, Ian Gilbert, MERCK SERENO, Beatriz Baragaña, Medicines for Malaria Venture, University of Dundee, Neil Norcross, 1469439-69-7, 1469439-71-1 , SUCCINATE
Fc1ccc2nc(cc(c2c1)C(=O)NCCN1CCCC1)-c1ccc(cc1)CN1CCOCC1