
The natural antioxidant resveratrol is detrimental for cardiovascular function when performing physical exercise
………………………..
………………….
Kiran Mazumdar-Shaw, chairman and managing director, Biocon.
Indian biopharma giant Biocon reported healthy growth of 22 percent for Q1 FY14 riding on the back of an increased geographical footprint in the emerging markets

Bangalore: Indian biopharma giant Biocon reported healthy growth of 22 percent for Q1 FY14. The firm clocked revenues worth $121 million (Rs723 crore), EBITDA of $29.50 million (Rs175 crore); and profit after tax (PAT) of $15.80 million (Rs94 crore).
Read more at: http://www.biospectrumasia.com/biospectrum/news/192549/how-biocon-clock-revenue-worth-usd121-mn#.UfdqX6I3CSo

Biocon’s India-focused branded formulations vertical as well as research services continue to grow at a steady pace


niraparib
MK-4827 Formula: C19H20N4O
MK-4827 Storage: at -20 ℃ 2 years
MK-4827 CAS No.: 1038915-60-4
MK-4827 is an inhibitor of PARP 1 and 2 with IC50 = 3.8 and 2.1 nM, respectively, currently in clinical trials. In a whole cell assay, it inhibited PARP activity with EC50 = 4 nM and inhibited proliferation of cancer cells with mutant BRCA-1 and BRCA-2 with CC50 in the 10-100 nM range. MK-4827 was well tolerated in vivo and demonstrated efficacy as a single agent in a xenograft model of BRCA-1 deficient cancer
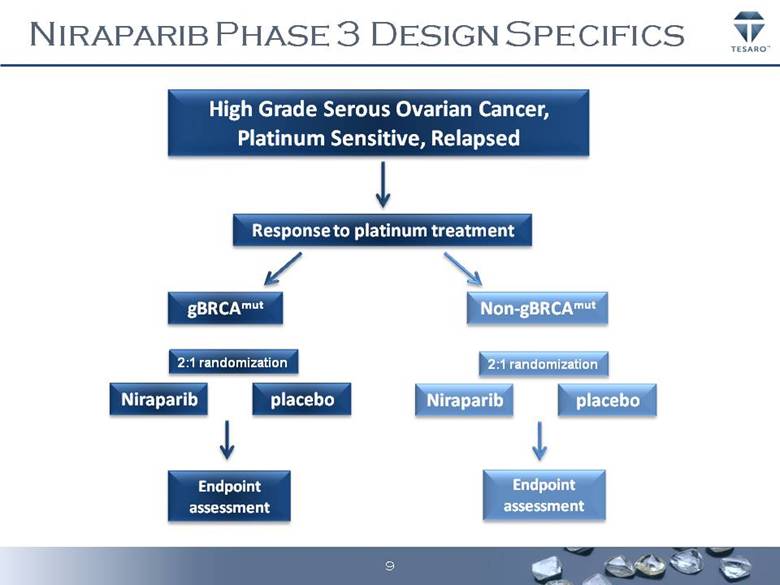
TESARO, Inc. an oncology-focused biopharmaceutical company, today announced that it has initiated patient enrollment in a Phase 3 trial of niraparib, an inhibitor of poly ADP-ribose polymerase (PARP), for the treatment of ovarian cancer. This trial, referred to as NOVA, will evaluate a single daily 300 milligram dose of niraparib in 360 patients with high grade serous, platinum sensitive, relapsed ovarian cancer compared to placebo.
Read more:
| Biological Activity of MK-4827: MK-4827 is a potent, selective, PARP 1/2 inhibitor with IC50 of 3.8 and 2.1 nM for PARP1 and 2, respectively. MK-4827 possesses potential antineoplastic activity. In a whole cell assay, MK-4827 prevented PARP activity with an EC50 of 4 nM, enhancing the accumulation of DNA strand breaks and promoting genomic instability and apoptosis. MK-4827 induces selective synthetic lethality in homologous recombination (HR) repair deficient tumors with BRCA1 / 2 loss and tumor cell lines with non-BRCA-related HR defects, supporting clinical utility in sporadic tumors. MK-4827 reveals good pharmacokinetic properties and is currently in phase I clin. trials. The phase I clinical trials for MK-4827 is ongoing in the treatment of solid tumors. |
| References on MK-4827: 1. Study of the Safety and Efficacy of MK-4827 Given With Temozolomide in Participants With Advanced Cancer (MK-4827-014 AM1).2. A Study of MK4827 in Participants With Advanced Solid Tumors or Hematologic Malignancies (MK-4827-001 AM8). 3. PARP inhibitor MK4827 4. Glendenning J, Tutt A. PARP inhibitors – Current Status and the Walk towards Early Breast cancer. Breast. 2011 Oct; 20 Suppl 3: S12-9. 5. Jones, Philip; Altamura, Sergio; Boueres, Julia et al. Discovery of 2 – {4 – [(3S)-Piperidin-3-yl] phenyl}-2H-INDAZOLE-7-carboxamide (MK-4827): A Novel Oral Poly (ADP-ribose) polymerase (PARP) Inhibitor efficacious in BRCA-1 and -2 Mutant Tumors. Journal of Medicinal Chemistry (2009), 52 (22), 7170-7185. |

DAPAGLIFLOZIN
AstraZeneca and Bristol-Myers Squibb Company today announced that the U.S. Food and Drug Administration (FDA) has acknowledged receipt of the New Drug Application (NDA) resubmission for investigational drug dapagliflozin for the treatment of adults with type 2 diabetes. The FDA assigned a new Prescription Drug User Fee Act goal date of January 11 2014.
The dapagliflozin Phase II/III clinical development program included more than 12,000 adult patients with diabetes (more than 8,000 patients received dapagliflozin) in 26 clinical trials. In response to the FDA’s January 2012 complete response letter requesting additional data to allow a better assessment of the benefit-risk profile of dapagliflozin, the NDA resubmission includes several new studies and additional long-term data (up to four years’ duration) from previously submitted studies, resulting in an overall increase in patient-years exposure to dapagliflozin of more than 50 percent.
Dapagliflozin, an investigational compound, is a selective and reversible inhibitor of sodium-glucose cotransporter 2 (SGLT2), which works independently of insulin. It is currently approved for the treatment of type 2diabetes in the European Union, Australia, Brazil, Mexico and New Zealand
READ ALL AT