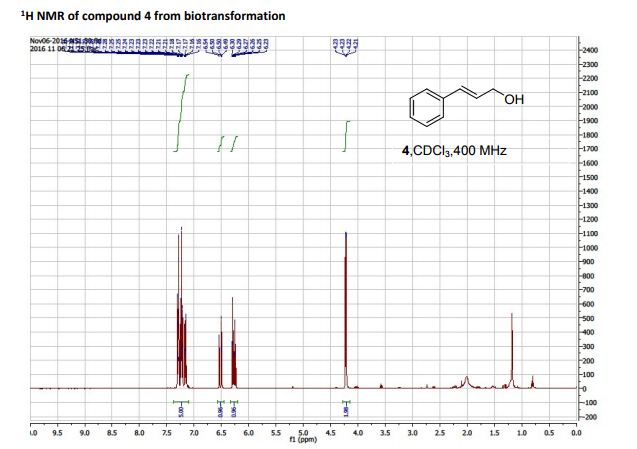
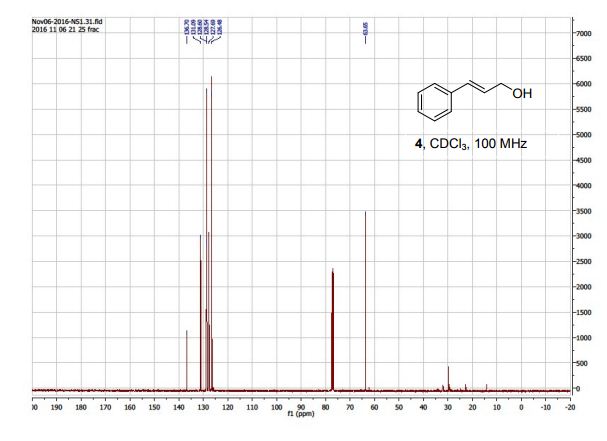
Tetrahydrothiopyran-4-one as Five-Carbon Source for Scalable Synthesis of (±)-Tapentadol
Ramagonolla Kranthikumar
- Prathama S. Mainkar
- Genji Sukumar
- Rambabu Chegondi
- Srivari Chandrasekhar*
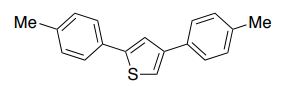
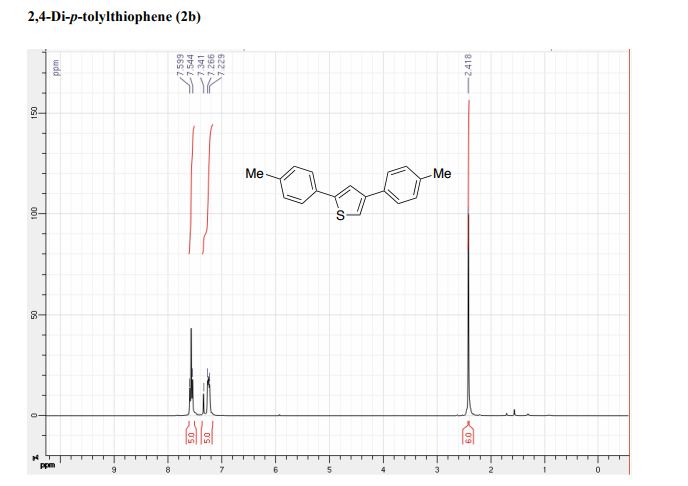
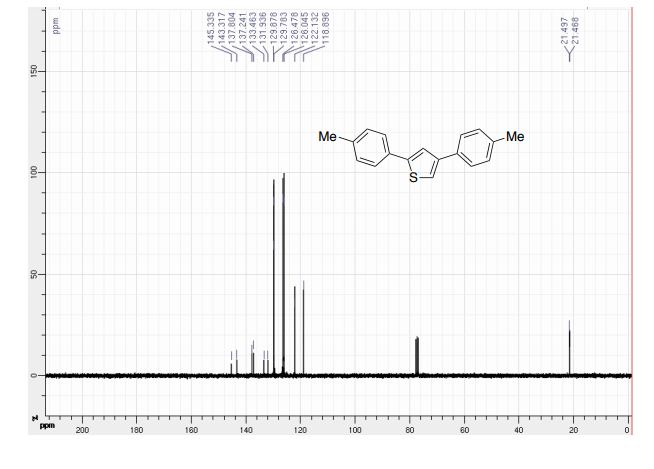
The improved process for the synthesis of (±)-tapentadol, the FDA-approved analgesic drug, is achieved from tetrahydrothiopyran-4-one as the five-carbon source.
////////























 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..




 +91- 999-997-2051
+91- 999-997-2051