
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Albany Molecular Research Inc. (AMRI) 26 Corporate Circle Albany, NY 12203 |



|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Albany Molecular Research Inc. (AMRI) 26 Corporate Circle Albany, NY 12203 |




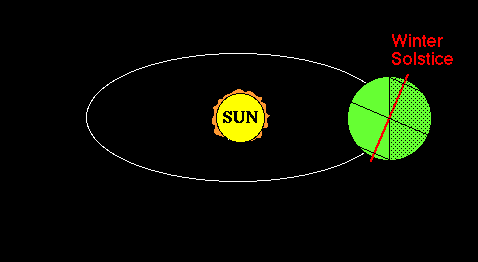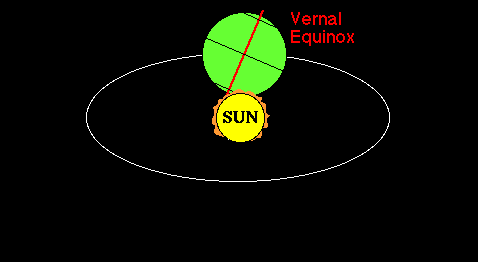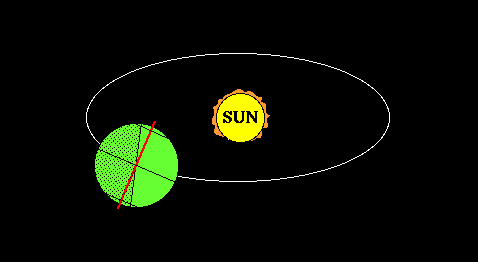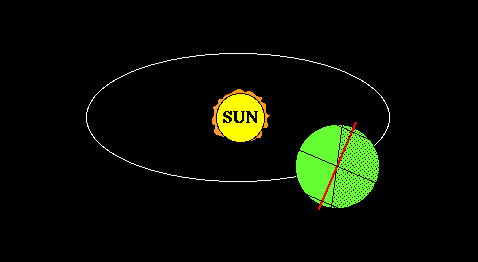
Lifitegrast, SAR 1118
SAR-1118-023
CAS 1025967-78-5
L-Phenylalanine, N-[[2-(6-benzofuranylcarbonyl)-5,7-dichloro-1,2,3,4-tetrahydro-6-isoquinolinyl]carbonyl]-3-(methylsulfonyl)-
INNOVATOR
SAR 1118 ophthalmic solution from SARcode Bioscience (Brisbane, Calif.) is a first-in-class molecule that inhibits T-cell inflammation by blocking the binding of two key cellular surface proteins (LFA-1 and ICAM-1) that mediate the chronic inflammatory cascade, so it may be able to reduce the inflammation associated with dry-eye disease.
A growing body of evidence points to a role for inflammation mediated by lymphocyte function-associated antigen-1 (LFA-1) and its ligand intercellular adhesion molecule-1 in the pathogenesis of diabetic macular oedema. This phase 1b clinical trial assessed the safety, tolerability, and pharmacokinetics of topically administered SAR 1118, a novel LFA-1 antagonist, in human subjects
Topical SAR 1118 was safe and well tolerated, and dose-dependent levels of drug were detected in aqueous. However, vitreous levels were below the threshold of detection with the concentrations tested. Further investigation of this medication for posterior segment applications would require intravitreal delivery or chemical modification of the drug.
In a Phase 2 dry eye trial, subjects receiving SAR 1118 demonstrated a reduction in corneal staining, increased tear production, and improved visual-related function as compared to placebo. These data were part of the scientific program of the Association for Research in Vision and Ophthalmology (ARVO) Annual Meeting held in Fort Lauderdale, Florida. SAR 1118 is a first-in-class topically administered small molecule integrin antagonist that inhibits T-cell mediated inflammation, a key component of dry eye disease.
In the randomized, placebo-controlled, multi-center trial, which included 230 subjects with dry eye disease, SAR 1118 demonstrated dose-dependent significant improvements (p<0.05) in inferior corneal staining over 12 weeks. As early as two weeks, a statistically significant(p<0.05) increase in tear production and improvement in visual-related functions (ability to read, drive at night, use a computer, and watch television) were observed, demonstrating early onset of action. Visual-related function was assessed using the Ocular Surface Disease Index (OSDI), a validated instrument designed to measure the severity of dry eye disease and the impact on vision-related function and quality of life. SAR 1118 was safe and well-tolerated with no serious ocular adverse events reported. Most ocular adverse events were transient and related to initial instillation.
“We are encouraged by the clinical effects of SAR 1118 in improving both signs and symptoms of dry eye, which supports the broad anti-inflammatory mechanism of this novel molecule,” commented Charles Semba, MD, Chief Medical Officer of SARcode Corporation. “We are excited to begin Phase 3 development in the later part of 2011, and have discussed appropriate and acceptable endpoints with the regulatory bodies to facilitate a smooth path towards approval.”
“It is well known that dry eye disease can have a deleterious effect on visual function, daily activities, workplace productivity, and quality of life. The potential to impact a patient’s quality of life in as early as 2 weeks could be a major advance in the current dry eye treatment model,” said Quinton Oswald, Chief Executive Officer of SARcode Corporation. “We hope to achieve similar results in our Phase 3 program.”
About Dry Eye Syndrome
Dry eye syndrome is a prevalent and often chronic condition estimated to affect approximately 20 million people in the US. It is among the most common diseases treated by ophthalmologists throughout the world, and has been shown to have a significant impact upon quality of life. Dry eye varies in severity and etiology, and symptoms most commonly manifest as discomfort, visual disturbances, and tear film instability due to decreased quality or quantity of tears. A major contributing factor towards the development of dry eye is inflammation caused by T-cell infiltration, proliferation and inflammatory cytokine production that can lead to reduction in tear film quality and ocular surface damage.
About SAR 1118 – SAR 1118 is a potent novel small molecule lymphocyte function-associated antigen-1 (LFA-1; CD11a/CD18; alphaLbeta2) antagonist under investigation for a broad range of ocular inflammatory conditions including dry eye and diabetic macular edema. LFA-1 is member of the integrin family of adhesion receptors found on the surface of all leukocytes and represents a therapeutic target central to a number of inflammatory stimuli. SAR 1118 has demonstrated potency in blocking LFA-1 binding to its cognate ligand, intercellular adhesion molecule-1 (ICAM-1; CD54), thereby inhibiting cell adhesion, cytokine production, and cellular proliferation in in vitro models.
About SARcode Corporation – SARcode Corporation, founded in 2006, is a venture-backed ophthalmic biopharmaceutical company based in Brisbane, CA. SARcode’s lead development program is a novel class of lymphocyte function-associated antigen-1 (LFA-1) antagonists for the treatment T-cell mediated inflammatory diseases. Institutional investors include Alta Partners and Clarus Venture Partners. For more information, visit www.sarcode.com
……………………….for a scheme see http://newdrugapprovals.org/2014/09/04/lifitegrast-sar-1118-effective-inhibitor-of-lfa-1-interactions-with-icam-1/
http://www.google.com.mx/patents/WO2005044817A1?cl=en
EXAMPLE 14 [0305] This example describes the synthesis of
[0306] which was prepared according to Scheme 9 and the procedure below.
[0307] SCHEME 9
[0308] a) To a solution of 3-carboxylbenzenesulfonyl chloride (3.54 g, 16 mmol) in ethyl acetate (50 mL) at 0 °C was added concentrated ammonia (2.5 mL). The reaction was neutralized with HCl in dioance (20 mL), diluted with ethyl acetate (100 mL), dried with anhydrous sodium sulfate and filtered. Concentration of the filtrate yielded the title compound, which was used without purification. [0309] b) Crude compound 14.1 was dissolved in THF (50 mL), to it was added borane (1.0 M in THF, 50 L) over 20 minute period. After the reaction was stirred at room temperature for 15 hours, the reaction was diluted with brine (20 mL) and water (10 mL), extracted with ethyl acetate (100 mL). The organic extract was dried over anhydrous sodium sulfate and filtered. Concentration of the filtrate yielded the title compound, which was used without further purification. [0310] c) To crude compound 14.2 solution in DCM (100 mL) was added activated 4A molecular sieve powder (8 g), pyridinium dichromate (7.55 g, 20 mmol). After the reaction was stirred at room temperature for 2 hours, the reaction mixture was filtered through silica gel (50 g), rinsed with ethyl acetate. The residue after concentration of the filtrate was purified by silca gel column with 30-50% ethyl acetate in hexane to give compound 14.3 (477mg, 16%, 3 steps). ESI-MS (m/z): (M+H4″) 186. [0311] d) Compound 14.4 was made according to Example 8e except that compound 14.3 was used instead of compound 8.7. MS (ESI4) m/z: 260 (M+H4″). [0312] e) Compound 14 was made according to Example 3g except that compound 14.4 was used instead of compound 3.4. 1H NMR (400 MHz, CD3OD) δ 7.89 (s, 1 H), 7.80 (s, 1 H), 7.75 (m, 2 H), 7.64 (s, 1 H), 7.57(d, 1 H), 7.34 (d, 2 H), 6.93 9s, 1 H), 5.00 (m, 1 H), 3.99 (m, 1 H), 3.73 (m, 1 H), 3.40 (dd, 1 H), 3.12 (dd, 1 H), 2.89 (m, 2 H) ppm; ESI-MS (m/z) 616 (M+H4″). [0313] EXAMPLE 15 [0314] This example describes the synthesis of
which was prepared according to Scheme 10 and the procedure below. [0315] SCHEME 10 rr–λ I BuLi, THF m-CPBA
s ) 2. DMF CH2CI2
15.1 15.2
[0316] a) To a solution of 0.2 mol of furan in 200 mL of dry THF was added 0.2 mol of «-BuLi (1.6 M in hexanes) at -78 °C, the resulting solution was stirred at room temperature for 4 hours. Subsequently, the mixture was cooled to -78 °C and treated with 0.21 mol of dimethyl disulfide, and the mixture was stirred at room temperature overnight, followed by adding 10 mL of saturated aqueous NH C1. The mixture was concentrated at room temperature, and the residue was diluted with 200 mL of saturated aqueous NH4C1 and extracted with ether. The extract was then washed with brine and dried with anhydrous Na2SO . The solvent was removed, and the residue was distilled to collect, the fraction at 135-140 °C/760 mmHg to give compound 15.1 in 55% yield. 1H NMR (400 MHz, CD3C1): δ 7.50 (s, IH), 6.45 (m, IH), 6.39 (s, IH), 2.42 (s, 3H) ppm. [0317] b) To a solution of 0.1 mol of compound 15.1 in 100 mL of dry THF was added 0.1 mol of n- uLi (1.6 M in hexanes) at -78 °C, the resulting solution was stirred at room temperature for 4 hours. Subsequently, the mixture was cooled to -78 °C and treated with 0.12 mol of dry DMF, and the mixture was stirred at room temperature overnight. The reaction was quenched by adding 10 mL of saturated aqueous NH4C1, and the mixture was concentrated. The residue was diluted with 100 mL of brine and extracted with EtOAC. The extract was washed with brine and dried with anhydrous Na2SO4. The solvent was removed and the residue was purified to give the title compound in 65% yield. 1H NMR (400 MHz, CD3C1): δ 9.52 (s, IH), 7.24 (d, J= 3.4 Hz, IH), 6.42 (d, J= 3.4Hz, IH), 2.60 (s, 3H) ppm; ESI-MS (m/z) (M+H4) 143.1. [0318] c) A mixture of 50 mmol of compound 15.2 and 120 mmol of -CPBA in 100 mL of CH2C12 was stirred at room temperature overnight. The mixture was diluted with 150 mL of CH2C12, and the mixture was washed with saturated aqueous NaHCO3 for several times. The solution was then dried with anhydrous Na2SO4 and concentrated. The residue was purified to give compound 15.3 in 70% yield. 1H NMR (400 MHz, CD3C1): δ 9.83 (s, IH), 7.33 (m, 2H), 3.27 (s, 3H) ppm; ESI-MS (m/z): (M+H4″) 175.0.
[0319] d) Compound 15.4 was made according to Example 8e except that compound 15.3 was used instead of 8.7. ESI-MS (m/z): (M+H4″) 248.1. [0320] e) Compound 15 was made according to Example except that compound 15.4 was used instead of 3.4. 1H NMR (400 MHz, CD3OD): δ 7.92 (s, IH), 7.76 (m, IH), 7.67 (s, IH), 7.34 (m, IH), 7.13 (s, IH), 6.69 (s, IH), 6.49 (s, IH), 5.11 (m, IH), 4.73 and 4.88 (m, 2H), 3.76 and 4.02 (m, 2H), 3.46 (m, IH), 3.30 (m, IH), 3.17 (s, 3H), 2.94 (m, 2H) ppm; ESI-MS (m/z): (M+H4) 605.05. [0321]
…………………………………….
US 20110092707
http://www.google.com/patents/US20110092707
Formula I:
has been found to be an effective inhibitor of Lymphocyte Function-Associated Antigen-1 (LFA-1) interactions with the family of Intercellular Adhesion Molecules (ICAM), and has desirable pharmacokinetic properties, including rapid systemic clearance. Improved forms, including crystalline forms, and their uses in treatment of disorders mediated by the interaction of LFA-1 and ICAM are described herein. Novel polymorphs of the compound of Formula I which may afford improved purity, stability, bioavailability and other like characteristics for use in pharmaceutical formulations and methods of use thereof are useful in treating disease.
Methods of Manufacture of the Compound of Formula I
In one embodiment, the compound of Formula I was synthesized as in the following Schemes 1-7. Alternate steps were used in the process as described below. The variants of this overall route yield superior yields, cost of goods and superior chiral purity compared to previously described methods. The final product of this synthesis yields crystalline Form A directly.
A first alternative protecting strategy produces compound 5, a trityl protected species as shown in Scheme 1. The synthesis begins by reductively aminating 3, 5, dichlorobenzaldehyde, compound 1, with 1-chloro-2-aminoethane and sodium cyanoborohydride in 35% yield. Cyclization of compound 2 using aluminum chloride catalysis and ammonium chloride at 185° C. provided compound 3 in 91% yield. Protection of the free amine of compound 3 as the trityl protected species afforded compound 4 in 89% yield. A carboxylic acid functionality was introduced by treatment of compound 4 with n-butyllithium (nBuLi) and Tetramethylethylenediamine (TMEDA), with subsequent introduction of carbon dioxide, to produce compound 5 in 75% yield.
Bromophenylalanine was used as the starting material for the right hand portion of the final molecule as shown in Scheme 2. t-Butylcarbamate (Boc) protection of the amino group was accomplished, using sodium bicarbonate (3 equivalents), t-butyl dicarbonate (Boc2O, 1.1 equivalent) in dioxane and water, to obtain compound 7 in 98% yield. A methyl sulfone functionality was introduced by treating the bromo compound 7 with copper iodide (0.4 equivalents), cesium carbonate (0.5 equivalents), L-proline (0.8 equivalents), and the sodium salt of methanesulfinic acid (3.9 equivalents) in dimethylsulfoxide (DMSO) at 95-100° C. for a total of 9 hours, with two further additions of copper iodide (0.2 equivalents) and L-proline (0.4 equivalents) during that period. Compound 8 was isolated in 96% yield. The carboxylic acid of compound 8 was converted to the benzyl ester, compound 9, in 99% yield, using benzyl alcohol (1.1 equivalent), dimethylaminopyridine (DMAP, 0.1 equivalent) and N-(3-dimethylaminopropyl)-N-ethylcarbodiimide (EDC, 1.0 equivalent). The amino group of compound 9 is deprotected by adding a 4N solution of HCl in dioxane to compound 9 at 0° C. in methylene chloride. The HCl salt of the free amino species, compound 10 was isolated in 94% yield.
Compound 5 was treated with triethylamine (TEA, 5 equivalents) and 2-(7-Aza-1H-benzotriazole-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate (HATU, 1.25 equivalents) for 10 minutes in dimethylformamide (DMF), and then compound 10 was added to the solution. After stirring at room temperature for 18 hours, the product, compound 11 was isolated in 70% yield. Removal of the trityl protecting group was accomplished by treating compound 1, with HCl in dioxane (4N, excess) at room temperature for 2 hours, diethyl ether added, and the solid product, compound 12, was isolated by filtration in 95% yield.
The benzofuranyl carbonyl moiety of the compound of Formula I was prepared using two alternative schemes, Scheme 4 and Scheme 4″. In one embodiment, the benzofuranyl carbonyl moiety was prepared by protecting the hydroxyl group of compound 13 by reacting with tert-butyldimethylsilyl chloride (1.0 equivalents) and triethylamine (TEA, 1.1 equivalents) in acetone, to give compound 14 in 79% yield. A solution of compound 14 in methanol was then treated with sodium borohydride (1.0 equivalent) at room temperature overnight. The reaction was quenched with an addition of acetone, stirred at room temperature for a further 2.5 hours, aqueous HCl (4N) was added with the temperature controlled to below 28C, tetrahydrofuran (THF) was added, and the solution stirred overnight under argon and in the absence of light. The product, compound 15, was isolated quantitatively by extraction into methylene chloride, concentrated at low heat, and used without further purification. The triflate ester, compound 16, was produced in 69% yield from compound 15 by reacting it with N-phenyl-bis(trifluoromethanesulfonimide) (1.0 equivalent) in methylene chloride for 72 hours. Compound 16 in a mixture of DMF, methanol, and triethylamine, was added to a prepared solution of palladium acetate, diphenyl, DMF and methanol in an autoclave. Carbon monoxide was charged into the autoclave to a pressure of 8 bar, and the reaction mixture was heated at 70° C. for 6 hours. After workup, compound 17 was isolated in 91% yield. Lithium hydroxide (4 equivalents) in methanol and water was used to hydrolyze the ester and permit the isolation of compound 18 in 97% yield.
In one embodiment, the benzofuranyl carbonyl moiety of the compound of Formula I was prepared according to Scheme 4″. By way of an Arbuzov reaction, diethyl 2-(1,3-dioxolan-2-yl)ethylphosphonate, compound 1″, was prepared from 2-(2-bromoethyl)-1,3-dioxolane by the addition of triethyl phosphate. After removal of ethyl bromide through distillation at 210° C. the crude reaction mixture was cooled and then by way of vacuum distillation, compound 1″ was collected as a colorless oil in 94% yield.
In the next step, n-butyllithium (2.15 equivalents) in hexane was cooled to −70° C. and diisopropylamine (2.25 equivalents) was added while keeping the temperature below −60° C. Compound 1″ (1 equivalent) dissolved in tetrahydrofuran (THF) was added over 30 min at −70° C. After 10 min, diethyl carbonate (1.05 equivalents) dissolved in THF was added over 30 min keeping the reaction temperature below −60° C. After stirring for one hour at −60° C., the reaction was allowed to warm to 15° C. and furan-2-carbaldehyde (1.3 equivalents) dissolved in THF was added. After stirring for 20 hrs at room temperature, the reaction was rotary evaporated to dryness to yield ethyl 2-(1,3-dioxolan2-yl)methyl-3-(furan-2-yl)acrylate, compound 5″. Crude compound 5″ was used directly in the next reaction.
The crude compound 5″ (1 equivalent) was dissolved in ethanol and added to a mixture of water and phosphoric acid (85%, 15 equivalents) over 30 min while keeping the temperature below 50° C. After stirring for 20 hrs at room temperature, another 200 ml of phosphoric acid (85%) was added and the mixture was heated to 50° C. for an additional two hrs. After removal of ethanol by rotary evaporation, the material was extracted with toluene, washed with water, dried with sodium sulfate, treated with charcoal, filtered and dried down to an oil. This oil was distilled to afford ethyl benzofuran-6-carboxylate, compound 6″, (bp 111-114.5° C.) which crystallized on standing. Compound 6″ was recovered at 57% yield based on compound 1″.
Compound 6″ (875 mmol) was dissolved in methanol and tetrahydrofuran (THF). Sodium hydroxide (4 M, 3 equivalents) was added and the reaction was stirred overnight. After concentration via rotary evaporation, the aqueous solution was extracted with methyl tert-butyl ether (MTBE), acidified to pH 2 with the addition of hydrochloric acid (HCl) and cooled resulting in fine crystals of benzofuran-6-carboxylic acid, i.e., compound 18. Compound 18 was isolated, washed with water and dried to a final yield of 97% yield.
The benzofuran carboxylic acid 18 was treated with oxalyl chloride (1.2 equivalents) and a catalytic amount of DMF, stirring for 5.5 hours until a clear solution was obtained. The solvent was removed under reduced pressure and the acid chloride of compound 18 was stored under argon until use, on the next day. The acid chloride, in methylene chloride was added slowly to a methylene chloride solution of the compound of Formula I and diisopropylethylamine (DIPEA) which was cooled to 0-5° C. The reaction was not permitted to rise above 5° C., and after completion of addition, was stirred at 5° C. for a further 0.5 hour. Upon aqueous workup and extraction with methylene chloride, the product, compound 19, was isolated in quantitative yield.
Taking the compound of Formula I directly as the crude reaction product after transfer hydrogenolysis, and reconcentrating down from a solution in methylene chloride, the amorphous form of the compound of Formula I was obtained in 97% purity.
An alternative protection strategy in this synthetic approach is illustrated in Scheme 6.
…………………….
WO 2014018748
http://www.google.com/patents/WO2014018748A1?cl=en
[0040] Methods of Manufacture of the Compound of Formula I

[0041] In one embodiment, the compound of Formula I is synthesized as in the following Schemes 1-7. The final product of this synthesis yields the compound of Formula I as an amorphous solid or as a crystalline form such as Forms A-E, or a pharmaceutically acceptable salt, either directly or indirectly. Variants of this overall route may provide superior yields, cost of goods, and/or superior chiral purity.
[0042] Protecting groups for amino and carboxy groups are known in the art. For example, see Greene, Protective Groups in Organic Synthesis, Wiley Interscience, 1981, and subsequent editions.
[0043] In various embodiments in the subsequent schemes, HATU is used as a reagent in amide- bond forming reactions. Alternatively, HATU is not used. In various embodiments, at least one amide-bond forming reaction is performed with thionyl chloride as a reagent in place of HATU. In various embodiments, all amide-bond forming reactions are performed with thionyl chloride as a reagent to form acid chlorides.
[0044] Scheme 1
[0045] A first alternative protecting strategy produces compound 5′, a protected species as shown in Scheme 1. The synthesis begins by reductively aminating 3, 5, dichlorobenzaldehyde, compound . Cyclization of compound 2′ provides compound 3′. Protection of the free amine of compound 3′ as a protected species provides compound 4′. A carboxylic acid functionality is introduced by treatment of compound 4′ with introduction of carbon dioxide, to produce compound 5′. In various embodiments, the protecting group of compound 4′ is a benzofuranyl carbonyl moiety derived from compound 18′.
[0046] In various embodiments, upon scaleup to multikilogram and larger scale reactions, treatment of compound 4′ with strong base (such as n-butyllithium (nBuLi) to generate a lithio species, or lithium diisopropyl amide (LDA) to generate the lithio species) is performed in flow mode rather than batchwise reaction due to instability of lithio species except at cold temperatures. Flow rates and residence times may be adjusted to maximize yield.
[0047] Scheme IB
5′ 4″”
[0048] In various embodiments, 6-hydroxy-l, 2,3, 4-tetrahydro-isoquino line (Compound 3″) is used as a starting material for Compound 5′. The starting material is chlorinated (x2) for example, with N-chlorosuccinimide. In various embodiments, the chlorination is performed in the presence of a sulfonic acid. In various embodiments, the sulfonic acid is selected from p- toluenesulfonic acid and methanesulfonic acid. Following protection of the amino group, the hydroxy group is functionalized, for example, as the triflate ester, which is carbonylated to yield the amino-protected methyl ester. Hydrolysis of the methyl ester yields the amino protected carboxylic acid.
[0049] Scheme 2
[0050] In various embodiments, bromophenyl alanine is used as the starting material for a portion of the final molecule as shown in Scheme 2. The starting material is protected with an amino protecting group to allow for introduction of a methyl sulfone functionality in compound 8′. Protecting groups are rearranged by introduction of an orthogonal protecting group for the carboxylic moiety, followed by deprotection of the amino group to provide compound 10′. In various embodiments, expensive or exotic bases are replaced with carbonate base such as potassium carbonate or calcium carbonate as a reagent.
[0051] Scheme 2A
10
[0052] In various embodiments, 3-methylsulfonylbenzaldehyde is converted into the 3- methylsulfonylphenylalanine derivative and functionalized to yield compound 10 as shown above.
[0053] Scheme 3
12′
[0054] Compounds 5′ and 10′ are joined through amide bond formation followed by deprotection of the remaining amino group in the presence of the carboxylic protecting group to yield compound 12′ or a salt thereof, such as the HCL salt.
[0055] Scheme 3
[0056] As an alternative to Scheme 3, compound 10″ is coupled with compound 5′ to yield the bromo compound 12″”, with subsequent introduction of a methyl sulfone functionality in place of bromine at a later step to produce compound 19′. Alternatively, instead of a bromine, compound 10″ includes X, where X is any halide (CI, I, Br, F) or a leaving group such as OTs, OTf, or the like.
[0057] Scheme 4
[0058] The benzofuranyl carbonyl moiety of the compound of Formula I can be prepared using various alternative schemes. In one embodiment, the benzofuranyl carbonyl moiety is prepared by protecting the hydroxyl group of compound 13′, reducing the carbonyl of compound 13′ to yield the benzofuranyl moiety, followed by carboxylation to yield compound 18′.
[0059] Scheme 4A
[0060] In one embodiment, compound 18′ is prepared from 6-hydroxybenzofuran via the triflate ester and the 6-carboxy methyl ester as intermediates, as shown in Example 4A.
[0061] Schem
[0062] The benzofuran carboxylic acid 18′ is coupled with compound 12′ (or a salt thereof) by amide bond formation to yield protected compound 19′, as shown in Scheme 5. Amide bond formation is known in the art
[0063] Schem
[0064] As an alternative to Schemes 3-5, compounds 18′ and 5″ may be coupled through amide bond formation followed by deprotection of the remaining carboxylic group to form compound 12″. Amide bond formation between compound 12″ and 10′ yields compound 19′ with a protected carboxylic group.
[0065] Scheme 5B
[0066] As an alternative to Schemes 1-5, compounds 12″ and 10″ may be coupled through amide bond formation followed by introduction of a methyl sulfone functionality in place of the bromine in converting compound 19″ to compound 19′ (similar to Scheme 2). Alternatively, instead of a bromine, compound 10″ includes X, where X is any halide (CI, I, Br, F) or a leaving group such as OTs, OTf, or the like. Compound 12″ can also be made using the following scheme:
[0067] Scheme 6
[0068] Final deprotection of compound 19′ to yield the compound of Formula I or a salt thereof is accomplished in a variety of ways. In various embodiments, the resulting compound of Formula I is provided in higher optical purity and/or higher overall purity and/or higher overall yield.
EXAMPLES
[00111] Example 1
Scheme El
[00112] Reductively aminating 3,5-dichlorobenzaldehyde, compound 1, with l-chloro-2- aminoethane and sodium cyanoborohydride provided 35% yield of compound 2. Cyclization of compound 2 using aluminum chloride catalysis and ammoniun chloride at 185°C provided compound 3 in 91% yield. Protection of the free amine of compound 3 as the trityl protected species afforded compound 4 in 89%> yield. A carboxylic acid functionality was introduced by treatment of compound 4 with n-butyllithium (nBuLi) and tetramethylethylenediamine (TMEDA), with subsequent introduction of carbon dioxide, to produce trityl protected compound 5 in 75% yield.
[00113] Example 1 A
2″
Scheme El A
[00114] To a glass reactor was charged 3,5-dichlorobenzaldehyde. Absolute ethanol was added to the batch slowly (this addition is mildly exothermic) and agitation started. 2,2- Diethoxyethyl amine (1.03 equiv) was slowly added to the batch, keeping the batch temperature at 20-78 °C. The batch was then heated to 76-78 °C for 2 h. GC-MS analysis indicated reaction completion (starting material < 1%). The batch was cooled to ambient temperature for work-up. The batch was concentrated in vacuo to a residue and azeotroped with heptanes (x2). The residue was cooled and held at 0-5 °C for 12 h to form a suspension. The solids were collected by filtration and the cake was washed with cold (0-5 °C) heptanes, and dried under hot nitrogen (45-50 °C) to afford Compound 2′ as a white solid (94% yield).
[00115] To a glass reactor was charged concentrated 95-98%) sulfuric acid (25.9 equiv).
The batch was heated to 120-125 °C and a solution of Compound 2′ in CH2CI2 was added slowly over 1 h, keeping the batch temperature between 120-125 °C. The batch was then stirred at 120— 125 °C for 6 h. The batch was cooled to < 50 °C. To a glass reactor was charged DI water and the batch temperature was adjusted to 0-5 °C. The reaction mixture was slowly transferred, keeping the batch temperature between 0-50 °C. DI water was used to aid the transfer. To the batch was added Dicalite 4200. The batch was filtered through a pad of Dicalite 4200. To the filtrate was added 50% aqueous sodium hydroxide solution slowly over 3 h, keeping the batch temperature between 0-50 °C to adjust the pH to 12. The resulting suspension was stirred at 45- 50 °C for 2 h and the solids were collected by filtration. The filter cake was slurried in DI water at 30-35 °C for 1 h. The batch was filtered. The cake was washed with heptanes and dried in vacuum oven at 45-50 °C for 22 h to give crude compound 2″ as a tan solid (75% yield), which was further purified by recrystallization.
[00116] To a reactor was added platinum dioxide (0.012 equiv), Compound 2″, and
MeOH (10 vol) and the suspension was stirred at room temperature under argon for 10 minutes. The reaction mixture was inerted with argon three times and then stirred under 125 psi of hydrogen at room temperature for 25 hours. HPLC analysis indicated complete reaction with less than 1% of the starting material remaining. After standing, the supernatant was decanted from the solids (catalyst) by vacuum. To the solids was added methanol and the slurry was mixed under nitrogen. The solids were allowed to settle on the bottom over several hours. The supernatant was decanted from the solids by vacuum. The combined supernatants were filtered through Celite under a blanket of nitrogen and the filter pad was washed with MeOH (x2). The combined filtrate and washes were concentrated to dryness. The residue was slurried in MTBE. The mixture was treated with 3 M HC1 while maintaining the temperature <40 °C resulting in the formation of a heavy precipitate. The mixture was stirred at 35-40 °C for 60 to 90 minutes. The batch was cooled to 0-5 °C, stirred for 60 to 90 minutes and then filtered. The filter cake was washed with cold DI water (x2) followed by a displacement wash with MTBE (x2). The filter cake was dried under reduced pressure to afford Compound 3 Hydrochloride Salt (86% yield). The hydrogenation catalyst can be recovered and re-used.
[00117] Compound 3 and trityl chloride were added to the reaction flask. DCM (10 vol) was added to the reactor and agitation was started to form slurry. The reaction mixture was cooled to 10-15 °C. N,N-Diisopropylethylamine (2.5 equiv) was slowly added to the reaction mixture, maintaining the temperature at 15-25 °C during the addition. Once addition was complete, the batch was stirred at 15 to 25 °C for a minimum of 60 minutes. The reaction was assayed by HPLC by diluting a sample with acetonitrile and then injecting it on the HPLC. The first assay after 30 minutes indicated that the reaction was complete with <1% of starting material observed by HPLC analysis. The reaction mixture was diluted with DI water (5 vol). The reaction mixture was stirred for 5 minutes after which it was transferred into a separation funnel and the phases were allowed to separate. The DCM layer was washed with DI water (5 vol) by stirring for 5 minutes and then allowing the phases to separate. The DCM layer was washed with brine (5 vol) by stirring for 5 minutes and then allowing the phases to separate. The DCM layer was dried over magnesium sulfate, filtered and the filter cake was washed with DCM (x2). The combined filtrate and washes were concentrated to a residue that was azeotroped with EtOAc (x2). The residue was suspended in EtOAc and stirred for 1 hour in a 40 °C water bath. The resulting slurry was cooled to 0-5 °C for 1 hour and then filtered. The filter cake was washed twice with EtOAc and then dried under reduced pressure to afford Compound 4.
[00118] Exam le IB
21 4″
[00119] To 1, 2,3, 4-tetrahydro-6-hydroxy-isoqino line in acetonitrile was added p- toluenesulfonic acid and N-chlorosuccinimide. The suspension was cooled to ambient temperature, and the product isolated by filtration for a yield of approximately 61% with purity greater than 95%. The isolated TsOH salt was recrystallized until purity was greater than 99.7%. To one equivalent of the TsOH salt suspended in methanol was added 2M sodium carbonate (0.55 eq.) and 1.2 eq. of Boc anhydride. The suspension was stirred at room temperature overnight. The reaction was monitored by HPLC. Upon completion, the mixture was cooled to below 10 °C, water was added, and the Boc-protected dichloro compound was isolated by filtraton. The product was washed and dried at 40 °C for a yield of 95% and purity of >97%. The Boc-protected dichloro compound was suspended in dichloromethane (10 volumes) and pyridine (5 volumes) was added. The mixture was cooled to below 2 °C, and triflic anhydride (1.25 eq) was added. The mixture was stirred at 0-2 °C for 10 minutes, and then poured into 10 volumes of 6%) aqueous sodium hydrogen carbonate solution. After washing with dichloromethane, the organic phases were combined and dried over magnesium sulphate. Following purification, the product (Compound 4′) was obtained in 90% yield and >98% purity. Compound 4′ was dissolved in dimethylformamide and methanol at room temperature. Diisopropylamine (4 eq) was added. Under CO atmosphere, l,3-bis(diphenylphosphino)propane (0.1 eq) and palladium acetate (0.1 eq) was added. The reaction was heated to refiux, and monitored by HPLC. Upon near completion, the mixture was cooled to ambient temperature. Workup with water, ethyl aceate, and brine yielded Compound 4″, which was used without further purification. Compound 4″ was dissolved in methanol and 2.4 M sodium hydroxide (10 volumes each) and refiuxed. The mixture was cooled to ambient temperature, and toluene was added. Following aqueous workup, the pH was adjusted to 2.3 with 3M hydrochloric acid, and crude product was isolated by filtration in 53% yield with greater than 80% purity.
[00120] Exam le 2
Scheme E2
[00121] t-Butylcarbamate (Boc) protection of the amino group of bromophenyl alanine was accomplished, using sodium bicarbonate (3 equivalents), t-butyl dicarbonate (Boc20, 1.1 equivalent) in dioxane and water, to obtain compound 7 in 98% yield. A methyl sulfone functionality was introduced by treating the bromo compound 7 with copper iodide (0.4 equivalents), cesium carbonate (0.5 equivalents), L-proline (0.8 equivalents), and the sodium salt of methanesulfinic acid (3.9 equivalents) in dimethylsulfoxide (DMSO) at 95-100°C for a total of 9 hours, with two further additions of copper iodide (0.2 equivalents) and L-proline (0.4 equivalents) during that period. Compound 8 was isolated in 96%> yield. The carboxylic acid of compound 8 was converted to the benzyl ester, compound 9, in 99% yield, using benzyl alcohol (1.1 equivalent), dimethylaminopyridine (DMAP, 0.1 equivalent) and N-(3- dimethylaminopropyl)-N-ethylcarbodiimide (EDC, 1.0 equivalent). The amino group of compound 9 is deprotected by adding a 4N solution of HC1 in dioxane to compound 9 at 0°C in methylene chloride. The HCl salt of the free amino species, compound 10 was isolated in 94% yield.
[00122] Example 2 A
[00123] Example 2 was repeated with potassium carbonate in place of cesium carbonate.
[00124] Example 2B
[00125] Boc-protected bromophenylalanine (Compound 7) (100g) was dissolved in
DMSO (400 mL) with stirring and degassing with argon. Sodium methane sulfmate (98g), copper iodide (28.7g), potassium carbonate (40 g) and L-proline (26.75g) were added at 28-30 °C. Reaction was heated to about 87 °C for about 17-19 hours. Reaction was cooled and quenched with crushed ice, stirred for 30-40 minutes, and the pH was adjusted from about 12 to about 3-4 with citric acid (350 g). Quenched reaction mixture was filtered, extracted with dichloromethane x3, washed with ammonium chloride solution, washed with sodium bisulphite solution, and washed with brine. Crude product in dichloromethane was concentrated in vacuo until moisture content was below about 0.5%, and used in next step without further isolation. Crude compound 8 in dichloromethane was charged with benzyl alcohol and DMPA with stirring under nitrogen. Reaction cooled to 0-5 °C. EDC-HCL (1.03 equiv) added with stirring for 30 minutes. Upon completion of reaction by TLC and HPLC, the reaction was quenched with sodium bicarbonate solution, the organic layer was separated, and the aqueous layer was extracted with dichloromethane. The organic layer was washed with citric acid solution, and combined organic layers were washed with brine solution. Dichloromethane was removed at 45- 50 °C, and the concentrate was used for next step without further isolation. The amino group of compound 9 was deprotected by adding a 4N solution of HCl in dioxane to compound 9 at 10- 15°C in methylene chloride. The HCl salt of the free amino species, compound 10 was isolated by filtration from diethyl ether. Isolation of compound 10 was performed through recrystallization using a dimethylformamide/dichloromethane solvent system.
[00126] Example 3
Scheme E3
[00127] Compound 5 was treated with triethylamine (TEA, 5 equivalents) and 2-(7-Aza- lH-benzotriazole-l-yl)-l,l,3,3-tetramethyluronium hexafluorophosphate (HATU, 1.25 equivalents) for 10 minutes in dimethylformamide (DMF), and then compound 10 was added to the solution. After stirring at room temperature for 18 hours, the product, compound 11 was isolated in 70% yield. Removal of the trityl protecting group was accomplished by treating compound 11, with HC1 in dioxane (4 N, excess) at room temperature for 2 hours, diethyl ether added, and the solid product, compound 12, was isolated by filtration in 95% yield. The compound 12 exists in both amorphous and crystalline form and can be isolated in either form.
[00128] Example 3 A
[00129] Compound 5 was dissolved in isopropyl acetate and cooled to 20 to 25 °C.
Thionyl chloride was added, with cooling to 10 to 15 °C, and N-methylmorpholine was added slowly. The reaction was monitored by HPLC. Compound 10, water, and isopropyl acetate were stirred at 15 to 20°C until a solution was achieved. N-methylmorpholine was added followed by addition of the Compound 5 reaction mixture (acid chloride of Compound 5). The reaction was monitored by HPLC. Upon completion, the biphasic layers were allowed to settle, and the aqueous layer was removed. The upper organic layer was extracted with water, and the remaining organic layer was distilled under vacuum. Dioxane and IpAc were added with further distillation. Once dry, 4N anhydrous HC1 in dioxane was added. The mixture was stirred at 20 to 25°C for 12 hours, and checked for complete deprotection by HPLC. Once complete, the thick slurry was filtered, washed with IP Ac and dried under vacuum at 45 to 55°C. Yield of Compound 12 was 88%.
[00130] Example 4
[00131] The benzofuranyl carbonyl moiety of the compound of Formula I was prepared using various schemes, (Schemes E4, E4A, and E4B).
15
Phenyl-bis-triflate
18 ‘
Scheme E4
[00132] The benzofuranyl carbonyl moiety was prepared by protecting the hydroxyl group of compound 13 by reacting with tert-butyldimethylsilyl chloride (1.0 equivalents) and triethylamine (TEA, 1.1 equivalents) in acetone, to give compound 14 in 79% yield. A solution of compound 14 in methanol was then treated with sodium borohydride (1.0 equivalent) at room temperature overnight. The reaction was quenched with an addition of acetone, stirred at room temperature for a further 2.5 hours, aqueous HCl (4N) was added with the temperature controlled to below 28 °C, tetrahydrofuran (THF) was added, and the solution stirred overnight under argon and in the absence of light. The product, compound 15, was isolated quantitatively by extraction into methylene chloride, concentrated at low heat, and used without further purification. The triflate ester, compound 16, was produced in 69% yield from compound 15 by reacting it with N- phenyl-bis(trifluoromethanesulfonimide) (1.0 equivalent) in methylene chloride for 72 hours. Compound 16 in a mixture of DMF, methanol, and triethylamine, was added to a prepared solution of palladium acetate, l,3-Bis(diphenylphosphino)propane (dppp), DMF and methanol in an autoclave. Carbon monoxide was charged into the autoclave to a pressure of 8 bar, and the reaction mixture was heated at 70 °C for 6 hours. After workup, compound 17 was isolated in 91% yield. Lithium hydroxide (4 equivalents) in methanol and water was used to hydro lyze the ester and permit the isolation of compound 18′ in 97% yield.
[00133] Example 4A
[00134] Example 4 was repeated with triflic anhydride and sodium hydroxide as reagents for the ester hydrolysis.
[00135] Compound 15 (6-Hydroxybenzofuran) was stirred in dichloromethane and diisopropylethylamine. Triflic anhydride (1.2 eq.) was added, keeping the temperature below 20C. The reaction was monitored by HPLC. The reaction was quenched with methanol, solvent was removed with vacuum, and the crude residue of Compound 16 was used without further purification. Compound 16 as crude residue was dissolved in 4 volumes of dimethylformamide and 2 volumes methanol. To the solution was added 0.02 eq. of palladium acetate, 0.02 eq. of dppp, and CO under pressure. The reaction was monitored by HPLC. Following workup, Compound 17 was isolated as a crude oily residue without further purification. The residue of compound 17 was dissolved in methanol (5 volumes) and 1 volume of sodium hydroxide (27.65%) was added. The mixture was heated to 40C until full conversion of HPLC. The mixture was cooled to ambient temperature and 3 volumes of water were added. The pH was adjusted to about 2 with 3M hydrochloric acid. The suspension was filtered, washed with water, and dried to give Compound 18’ in about 75% overall yield with purity >99.5%.
[00136] Example 4B
Scheme E4B [00137] Diethyl 2-(l,3-dioxolan-2-yl)ethylphosphonate, compound 1″, was prepared from
2-(2-bromoethyl)-l,3-dioxolane by the addition of triethyl phosphate. After removal of ethyl bromide through distillation at 210°C the crude reaction mixture was cooled and then by way of vacuum distillation, compound 1″ was collected as a colorless oil in 94% yield.
[00138] In the next step, n-butyllithium (2.15 equivalents) in hexane was cooled to -70 °C and diisopropylamine (2.25 equivalents) was added while keeping the temperature below -60 °C. Compound 1″ (1 equivalent) dissolved in tetrahydrofuran (THF) was added over 30 min at -70 °C. After 10 min, diethyl carbonate (1.05 equivalents) dissolved in THF was added over 30 min keeping the reaction temperature below -60 °C. After stirring for one hour at -60 °C, the reaction was allowed to warm to 15 °C and furan-2-carbaldehyde (1.3 equivalents) dissolved in THF was added. After stirring for 20 hrs at room temperature, the reaction was rotary evaporated to dryness to yield ethyl 2-((l,3-dioxolan2-yl)methyl-3-(furan-2-yl)acrylate, which was used directly in the next reaction.
[00139] The crude compound (1 equivalent) was dissolved in ethanol and added to a mixture of water and phosphoric acid (85%>, 15 equivalents) over 30 min while keeping the temperature below 50°C. After stirring for 20 hrs at room temperature, another 200 ml of phosphoric acid (85%>) was added and the mixture was heated to 50 °C for an additional two hrs.
After removal of ethanol by rotary evaporation, the material was extacted with toluene, washed with water, dried with sodium sulfate, treated with charcoal, filtered and dried down to an oil. This oil was distilled to afford ethyl benzofuran-6-carboxylate, compound 6″, (bp 111-114.5°C) which crystallized on standing. Compound 6″ was recovered at 57%> yield based on compound
1″.
[00140] Compound 6″ (875 mmol) was dissolved in methanol and tetrahydrofuran (THF).
Sodium hydroxide (4 M, 3 equivalents) was added and the reaction was stirred overnight. After concentration via rotary evaporation, the aqueous solution was extracted with methyl tert-butyl ether (MTBE), acidified to pH 2 with the addition of hydrochloric acid (HC1) and cooled resulting in fine crystals of benzofuran-6-carboxylic acid, i.e., compound 18′. Compound 18′ was isolated, washed with water and dried to a final yield of 97%> yield.
[00141] Example 5
10% Pd/C, HCOOH/NEt3
MeOH/THF 5:1
Form A of Formula I
Scheme E5
[00142] The benzofuran carboxylic acid 18′ was treated with oxalyl chloride (1.2 equivalents) and a catalytic amount of DMF, stirring for 5.5 hours until a clear solution was obtained. The solvent was removed under reduced pressure and the acid chloride of compound 18′ was stored under argon until use, on the next day. The acid chloride, in methylene chloride was added slowly to a methylene chloride solution of the compound of Formula 12 and diisopropylethylamine (DIPEA) which was cooled to 0-5 °C. The reaction was not permitted to rise above 5°C, and after completion of addition, was stirred at 5°C for a further 0.5 hour. Upon aqueous workup and extraction with methylene chloride, the product, compound 19, was isolated in quantitative yield.
[00143] The benzyl ester of compound 19 was removed by transfer hydrogenolysis using
10% palladium on carbon, using formic acid and triethylamine in a 5: 1 mixture of methanol:THF, to produce the compound of Formula I in 95% yield.
[00144] A final step of slurrying in methyl ethylketone (MEK) produced Form A of the compound of Formula I. The product was washed with water to remove residual MEK. Alternatively, the product of the hydrogenolysis step was slurried in acetonitrile to yield Form A of the compound of Formula I.
[00145] Taking the compound of Formula I directly as the crude reaction product after transfer hydrogenolysis, and reconcentrating down from a solution in methylene chloride, the amorphous form of the compound of Formula I was obtained in 97% purity.
[00146] Example 6
[00147] An alternative protection strategy was performed in Scheme E6.
Scheme E6
[00148] Boc-protection was used for the ring nitrogen in the intermediates 21 and 22.
Compound 5 was deprotected with HC1 in dioxane to produce compound 23 in better than 97%> yield. Boc-protection was introduced, using di-tert-butyl dicarbonate (1.1 equivalent), and compound 21 was obtained in better than 95% yield. Compound 10 was coupled with compound 21 to obtain compound 22, using HATU and triethylamine in DMF. The product, compound 22, was obtained in quantitative yield, and greater than 90% purity. Deprotection with HC1 yielded the compound of Formula 12 in 97.4% yield.
[00149] Transfer hydrogeno lysis of compound 19 produced the compound of Formula I with optical purity of 98.5% (S) enantiomer compared to 79-94.5% (S) enantiomer optical purity obtained by hydrolysis of the corresponding methyl ester.
……………………………..

LFA-1/ICAM-1 interaction is essential in support of inflammatory and specific T-cell regulated immune responses by mediating cell adhesion, leukocyte extravasation, migration, antigen presentation, formation of immunological synapse, and augmentation of T-cell receptor signaling. The increase of ICAM-1 expression levels in conjunctival epithelial cells and acinar cells was observed in animal models and patients diagnosed with dry eye. Therefore, it has been hypothesized that small molecule LFA-1/ICAM-1 antagonists could be an effective topical treatment for dry eye. In this letter, we describe the discovery of a potent tetrahydroisoquinoline (THIQ)-derived LFA-1/ICAM-1 antagonist (SAR 1118) and its development as an ophthalmic solution for treating dry eye.
http://pubs.acs.org/doi/suppl/10.1021/ml2002482/suppl_file/ml2002482_si_001.pdf
| Cited Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US8084047 * | Jul 23, 2009 | Dec 27, 2011 | Sarcode Bioscience Inc. | Compositions and methods for treatment of eye disorders |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US8367701 | Nov 4, 2011 | Feb 5, 2013 | Sarcode Bioscience Inc. | Crystalline pharmaceutical and methods of preparation and use thereof |
| US8592450 | Feb 16, 2012 | Nov 26, 2013 | Sarcode Bioscience Inc. | Compositions and methods for treatment of eye disorders |
| US8758776 | Jan 21, 2011 | Jun 24, 2014 | Sarcode Bioscience Inc. | Compositions and methods for treatment |
| US8771715 | Jan 21, 2011 | Jul 8, 2014 | Sarcode Bioscience Inc. | Compositions and methods for treatment |
| WO2012121659A1 * | Mar 8, 2012 | Sep 13, 2012 | Kat2Biz Ab C/O Interpares Konsult Ab | Reduction of c-0 bonds by catalytic transfer hydrogenolysis |
| WO2014018748A1 * | Jul 25, 2013 | Jan 30, 2014 | Sarcode Bioscience Inc. | Lfa-1 inhibitor and polymorph thereof |



The journey of a pharmaceutical product
In every process of the drug development, from manufacturing to shipping and transportation after shipments, there are strict standards in place, ranging from those defined by the Pharmaceutical Affairs Law, those that require approval from regulatory agencies, and unique standards set within companies.

……………………………………………………………………………….



Researchers from the Nagasaki University in Japan have developed a new method to detect the presence of Ebola virus in 30 minutes.
The new method is claimed to allow doctors to rapidly diagnose the infection.
Professor Jiro Yasuda and team was quoted by AFP as saying that the newly developed process is cheaper than the system, which is currently in use in West Africa where the virus has already claimed around 1,500 lives.
Yasuda said: “The new method is simpler than the current one and can be used in countries where expensive testing equipment is not available.
Japanese researchers develop new 30-minute method to detect Ebola virus
http://www.pharmaceutical-technology.com/news/newsjapanese-researchers-develop-new-30-minute-method-detect-ebola-virus-4360875?WT.mc_id=DN_News
Researchers from the Nagasaki University in Japan have developed a new method to detect the presence of Ebola virus in 30 minutes.
Tokyo (AFP) – Japanese researchers said Tuesday they had developed a new method to detect the presence of the Ebola virus in 30 minutes, with technology that could allow doctors to quickly diagnose infection.
Professor Jiro Yasuda and his team at Nagasaki University say their process is also cheaper than the system currently in use in west Africa where the virus has already killed more than 1,500 people.
“The new method is simpler than the current one and can be used in countries where expensive testing equipment is not available,” Yasuda told AFP by telephone.
“We have yet to receive any questions or requests, but we are pleased to offer the system, which is ready to go,” he said.
Yasuda said the team had developed what he called a “primer”, which amplifies only those genes specific to the Ebola virus found in a blood sample or other bodily fluid.
Using existing techniques, ribonucleic acid (RNA) — biological molecules used in the coding of genes — is extracted from any viruses present in a blood sample.
This is then used to synthesise the viral DNA, which can be mixed with the primers and then heated to 60-65 degrees Celsius (140-149 Fahrenheit).
If Ebola is present, DNA specific to the virus is amplified in 30 minutes due to the action of the primers. The by-products from the process cause the liquid to become cloudy, providing visual confirmation, Yasuda said.
Currently, a method called polymerase chain reaction, or PCR, is widely used to detect the Ebola virus, which requires doctors to heat and cool samples repeatedly and takes up to two hours.
“The new method only needs a small, battery-powered warmer and the entire system costs just tens of thousands of yen (hundreds of dollars), which developing countries should be able to afford,” he added.
The outbreak of the Ebola virus, transmitted through contact with infected bodily fluids, has sparked alarm throughout western Africa and further afield.
JAPAN

Idasanutlin(RG-7388)
cas 1229705-06-9
4-{ [(2R,3S,4R,5S)-4-(4-Chloro-2-fluoro-phenyl)-3-(3-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2- dimethyl-prop yl)-pyrrolidine-2-carbonyl] -amino }-3-methoxy-benzoic acid (C31H29Cl2F2N304)
4-{[(2R,3S,4R,5S)-4-(4-Chloro-2-fluoro-phenyl)-3-(3-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carbonyl]-amino}-3-methoxy-benzoic acid
MW 616.4973
F. Hoffmann-La Roche Ag, Hoffmann-La Roche Inc.ROCHE PHASE1
for the oral treatment of cancer, including solid tumors and hematological tumors, including acute myelogenous leukemia
Acute myelogenous leukemia; Cancer; Prostate tumor
Mdm2 p53-binding protein inhibitor
RG7388 is the second generation inhibitor of P53-MDM2 interaction. It is orally active, potently and selectively antagonizing the P53-MDM2 interaction with Ki at low nM. It is designed to selectively target MDM2, a key negative regulator of the p53 tumor suppressor protein. Blocking this essential interaction may lead to apoptosis via activation of p53 in tumor cells with functional p53 signaling. It is currently in clinical evaluation.
Description:
Value IC50: 30 nM (IC50 Average of three wt-p53 SJSA1 Cancer cell lines, RKO, HCT116)
. RG7388 is an Oral, Selective, small molecule antagonist that inhibits binding of MDM2 to p53 MDM2 Blocking the MDM2-p53 Interaction stabilizes p53 and activates p53-mediated cell death and inhibition of cell Growth.
RG7388 Showed all the Characteristics expected of an MDM2 inhibitor in terms of speci? c binding to the target, mechanistic outcomes Resulting from Activation of the p53 pathway, and in vivo ?. Although e cacy Mechanism of Action of the cellular is identical to that of RG7388 RG7112, it is much More potent and Selective.
Tumor suppressor p53 is a powerful growth suppressive and pro-apoptotic protein that plays a central role in protection from tumor development.A potent transcription factor, p53 is activated following cellular stress and regulates multiple downstream genes implicated in cell cycle control, apoptosis, DNA repair, and senescence.While p53 is inactivated in about 50% of human cancers by mutation or deletion, it remains wild-type in the remaining cases but its function is impaired by other mechanisms. One such mechanism is the overproduction of MDM2, the primary negative regulator of p53, which effectively disables p53 function.An E3 ligase, MDM2 binds p53 and regulates p53 protein levels through an autoregulatory feedback loop. Stabilization and activation of wild-type p53 by inhibition of MDM2 binding has been explored as a novel approach for cancer therapy.
……………………………..

Restoration of p53 activity by inhibition of the p53–MDM2 interaction has been considered an attractive approach for cancer treatment. However, the hydrophobic protein–protein interaction surface represents a significant challenge for the development of small-molecule inhibitors with desirable pharmacological profiles. RG7112 was the first small-molecule p53–MDM2 inhibitor in clinical development. Here, we report the discovery and characterization of a second generation clinical MDM2 inhibitor, RG7388, with superior potency and selectivity.
Restoration of p53 activity by inhibition of the p53–MDM2 interaction has been considered an attractive approach for cancer treatment. However, the hydrophobic protein–protein interaction surface represents a significant challenge for the development of small-molecule inhibitors with desirable pharmacological profiles. RG7112 was the first small-molecule p53–MDM2 inhibitor in clinical development. Here, we report the discovery and characterization of a second generation clinical MDM2 inhibitor, RG7388, with superior potency and selectivity.
Restoration of p53 activity by inhibition of the p53–MDM2 interaction has been considered an attractive approach for cancer treatment. However, the hydrophobic protein–protein interaction surface represents a significant challenge for the development of small-molecule inhibitors with desirable pharmacological profiles. RG7112 was the first small-molecule p53–MDM2 inhibitor in clinical development. Here, we report the discovery and characterization of a second generation clinical MDM2 inhibitor, RG7388, with superior potency and selectivity.
compd 12
1H NMR (400 MHz, DMSO-d6)
δ12.86 (s, 1 H), 10.46 (s, 1 H), 8.35 (d, J = 8.86 Hz, 1 H), 7.71 (t, J = 6.95 Hz, 1 H), 7.48 – 7.61 (m,4 H), 7.29 – 7.42 (m, 3 H), 4.53 – 4.61 (m, 2 H), 4.38 (br. s., 1 H), 3.86 – 3.99 (m, 4 H), 1.62 (dd,J = 9.87, 14.00 Hz, 1 H), 1.24 (d, J = 14.00 Hz, 1 H), 0.95 (s, 9 H) ppm;
13C NMR (101 MHz, DMSO-d6) δ 171.2, 166.9, 160.8, 158.3, 156.8, 154.4, 147.5, 134.8, 134.7, 131.0, 130.8, 130.0,
128.6, 126.1, 125.9, 125.6, 125.3, 122.7, 119.6, 119.4, 119.2, 119.1, 117.7, 117.4, 117.3, 117.2, 111.0,
64.7, 63.4, 63.3, 63.3, 63.2, 55.8, 50.2, 43.9, 30.1, 29.5, 25.5 ppm;
HRMS (ES+) m/z CalcdC31H29Cl2F2N3O3+ H [M+H]+: 616.1576, found: 616.1574.
Anal. Calcd for C31H29Cl2F2N3O3: C, 60.4; H, 4.74; Cl, 11.5; F, 6.16; N, 6.82. Found: C, 60.3; H, 4.79; Cl, 11.3; F, 6.02; N, 6.82.
…………………………….
see
WO-2014128094
http://www.google.com/patents/WO2014128094A1?cl=en
F Hoffmann-La Roche AG; Hoffmann-La Roche Inc
Asymmetric synthesis of a substituted pyrrolidine-2-carboxamide
Process for the preparation of RG-7388 and their novel intermediates. Roche is developing idasanutlin (RG-7388), a small-molecule MDM2 antagonist that inhibits binding of MDM2 to p53, for the oral treatment of cancer, including solid tumors and hematological tumors, including acute myelogenous leukemia, as of September 2014, the drug is in Phase 1 trials. See WO2014114575 claims physically stable solid dispersion comprising a compound eg idasanutlin, with an aqueous solubility of less than 1 μg/ml and an ionic polymer eg copovidone, for treating cancer.
Compound I.
Scheme 2
process to produce a compound of the formula
which comprises
a) reacting a compound of the formula (IV)
with a compound of the formula (V)
in the presence of a silver catalyst; b) isomerising the product of (a) by reaction with a suitable base selected from a strong amine or with an insoluble base in the above solvents at a temperature range of from about 20 to 80 °C; and c) hydrolyzing the product of (b) in any suitable hydroxide in a solvent having water miscibility at a temperature between about 20 to about 80°C to obtain a compound of formula I; wherein
R1 is a non-tertiary alkyl or benzyl, or other ester protecting group.
Example 1 : (Z)-3-(3-Chloro-2-fluoro-phenyl)-2-(4-chloro-2-fluoro-phenvl)-acrvlonitrile
A 250-L glass-lined reactor was charged with 2-(4-chloro-2-fluorophenyl)acetonitrile (15.0 kg, 88.5 mol, Eq: 0.988), 3-chloro-2-fluorobenzaldehyde (14.2 kg, 89.6 mol, Eq: 1.00), MeOH (140 L). In one portion, a solution of sodium hydroxide [prepared from 50 wt% solution (0.23 L, 4.4 mmol, Eq: 0.05) diluted in methanol (10 L)] was added. The resulting mixture was heated to 50 °C for 4.5 h, and then the resulting thick slurry was cooled down to 20 °C. Consumption of 3- chloro-2-fluorobenzaldehyde was monitored by HPLC analysis. The solid product was isolated by filtration via a 0.3 m filter/dryer and the cake washed with methanol (58 L). The product was dried under vacuum with N2 purge at 60°C to provide the stilbene as a white powder, 24.2 kg (88.5% yield) with 99.87% purity by HPLC analysis.
1H NMR (300 MHz, CDC13) δ 8.10-8.15 (1H, m), 7.79 (1H, s), 7.48-7.59 (2H, m), 7.20-7.28 (3H, m).
Compound 5: 1H NMR (400 MHz, DMSO-d6) δ: 12.89 (br. s., 1H), 10.50 (s, 1H), 8.39 (d, J = 8.8 Hz, 1H), 7.75 (t, J = 6.8 Hz, 1H), 7.51 – 7.64 (m, 4H), 7.33 – 7.46 (m, 3H), 4.57 – 4.66 (m, 2H), 4.36 – 4.47 (m, 1H), 3.95 – 4.03 (m, 1H), 3.94 (s, 3H), 1.66 (dd, J = 14.2, 9.9 Hz, 1H), 1.28 (d, J = 13.8 Hz, 1H), 0.99 (s, 9H).
A 500-mL, round bottomed flask equipped with a magnetic stirrer and nitrogen inlet/bubbler was charged with copper(II) acetate (150 mg, 0.826 mmol), (R)-BINAP (560 mg, 0.899 mmol), and 2-methyltetrahydrofuran (120 mL). The suspension was stirred at room temperature under N2 for 3 h when a clear blue solution was obtained. Then 12.0 mL (68.7 mmol) of N,N- diisopropylethylamine was added, followed by 20.0 g (64.5 mmol) of Compound (1) and 24.0 g (71.8 mmol) of Compound (2). The suspension was stirred at room temperature under N2 for 18 h, and LCMS analysis indicated complete reaction. The reaction mixture was diluted with 100 mL of 5% ammonium acetate solution and stirred for 15 min, then poured into a 500-mL separatory funnel. The organic phase separated was washed with an additional 5% ammonium acetate solution (100 mL), then with 100 mL of 5% sodium chloride solution (100 mL), and „
– 27 – concentrated at 40 °C under reduced pressure to a thick syrup (ca. 60 g ). This syrup (containing 6 and 7) was dissolved in tetrahydrofuran (120 mL), methanol (60.0 mL), and water (6.00 mL). Then sodium hydroxide (50% solution, 6.00 mL, 114 mmol) was added dropwise. The mixture was stirred at room temperature for 18 h. LCMS and chiral HPLC indicated complete hydrolysis and isomerization. The reaction mixture was acidified with 20.0 mL (349 mmol) of acetic acid, and then concentrated at 40 °C under reduced pressure to remove ca. 80 mL of solvent. The residue was diluted with 2-propanol (200 mL), and further concentrated to remove ca. 60 mL of solvent, and then water (120 mL) was added. The slurry was stirred under reflux for 1 h, at room temperature overnight, then filtered and the flask was rinsed with of 2-propanol- water (2: 1) (20.0 mL). The filter cake was washed with 2-propanol- water (1: 1) (2 x 100 mL = 200 mL), and with water (2 x 200 mL = 400 mL), then vacuum oven dried at 60 °C to give 33.48 g (84.2% yield) of crude Compound 5 as a white solid ; 99.26% pure and 87.93% ee as judged by LCMS and chiral HPLC analysis. Compound 6 (exo cycloaddition product, 2,5-cis): 1H NMR (400 MHz, CDC13) δ 9.66 (brs, 1H),
8.42 (d, J = 8.3 Hz, 1H), 7.89 (m, 1H), 7.65 (dd, J = 8.6, 1.8 Hz, 1H), 7.55 (d, J = 1.8 Hz, 1H), 7.40 (m, 1H), 7.32 (td, J = 8.3, 1.5 Hz, 1H), 7.22-7.15 (m, 3H), 4.45 (m, 2H), 4.36 (q, J = 7.2 Hz, 2H), 4.25 (m, 1H), 3.91 (s, 3H), 1.39 (t, J = 7.2 Hz, 3H), 1.30 (dd, J = 14.2, 9.3 Hz, 1H), 0.92 (s, 9H), 0.84 (d, J = 14.2 Hz, 1H).
Compound 7 (endo cycloaddition product, 2,5-cis): 1H NMR (400 MHz, CDC13) δ 9.97 (brs, 1H), 8.30 (d, J = 8.4 Hz, 1H), 7.65 (dd, J = 8.3, 1.8 Hz, 1H), 7.56 (d, J = 1.7 Hz, 1H), 7.51 (m, 1H),
7.43 (t, J = 8.4 Hz, 1H), 7.23 (m, 1H), 7.17 (dd, J =12.6, 2.0 Hz, 1H), 7.11 (m, 1H), 6.89 (td, J = 8.1, 1.2 Hz, 1H), 5.05 (dd, J = 10.8, 2.1 Hz, 1H), 4.53 (d, J = 10.8 Hz, 1H), 4.37 (q, J = 7.2 Hz, 2H), 4.22 (d, J = 8.7 Hz, 1H), 3.95 (s, 3H), 1.85 (dd, J = 14.1, 8.7 Hz, 1H), 1.48 (d, J =14.1 Hz, 1H), 1.40 (t, J = 7.2 Hz, 1H), 0.97 (s, 9H).
…………………
http://www.google.com/patents/WO2014114575A1?cl=en
The compound 4-{ [(2R,3S,4R,5S)-4- (4-Chloro-2-fluoro-phenyl)-3-(3-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)- pyrrolidine-2-carbonyl] -amino }-3-methoxy-benzoic acid (Compound A), as well as methods for making it, is disclosed in U.S. Patent No. 8,354,444 and WO2011/098398.

4-{ [(2R,3S,4R,5S)-4-(4-Chloro-2-fluoro-phenyl)-3-(3-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2- dimethyl-prop yl)-pyrrolidine-2-carbonyl] -amino }-3-methoxy-benzoic acid (C31H29Cl2F2N304) (Compound A) is a potent and selective inhibitor of the p53-MDM2 interaction that activates the p53 pathway and induces cell cycle arrest and/or apoptosis in a variety of tumor types expressing wild-type p53 in vitro and in vivo. Compound A belongs to a novel class of MDM2 inhibitors having potent anti-cancer therapeutic activity, in particular in leukemia such as AML and solid tumors such as for example non-small cell lung, breast and colorectal cancers.
The above-identified international patent application and US Patent describe Compound A in crystalline form and is herein incorporated by reference in its totality. The crystalline form of the compound has an on- set melting point of approximately 277 °C. The crystalline forms have relatively low aqueous solubility (<0.05 μg/mL in water) at physiological pHs (which range from pHl.5-8.0) and consequently less than optimal bioavailability (high variability)
…………………………..
Compound A is an orally administered pyrrolidine that inhibits the binding of MDM2 to p53 and is thus useful in the treatment of cancer. It has the following chemical structure:
Molecular Weight =616.4973
Molecular Formula =C31 H29CI2F2N304
Compound A recently entered into phase I clinical trials for the treatment of solid tumors. See ClinicalTrials.gov, identifier NCT01462175. This compound is disclosed in US Pub 2010/0152190 A1 . To the extent necessary, this patent publication is herein incorporated by reference. The Compound A, as well as a method for making it, is also disclosed in WO201 1/098398.
Applicants have discovered that Compound A is especially effective, and best tolerated, in cancer therapy when administered in the specific doses and pursuant to the specific protocols herein described.
………………………..
http://www.google.com/patents/US20100152190
Example 52a Preparation of intermediate (Z)-3-(3-chloro-2-fluoro-phenyl)-2-(4-chloro-2-fluoro-phenyl)-acrylonitrile
In a manner similar to the method described in Example 1b, 4-chloro-2-fluorophenylacetonitrile (5 g, 30 mmol) was reacted with 3-chloro-2-fluorobenzaldehyde (5 g, 32 mmol), methanolic solution (25 wt %) of sodium methoxide (21 mL, 92 mmol) in methanol (200 mL) at 45° C. for 5 h to give (Z)-3-(3-chloro-2-fluoro-phenyl)-2-(4-chloro-2-fluoro-phenyl)-acrylonitrile as a white powder (9 g, 97%).
Example 52b Preparation of intermediate rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid tert-butyl ester
In a manner similar to the method described in Example 1c, [3-methyl-but-(E)-ylideneamino]-acetic acid tert-butyl ester prepared in Example 1a (2.3 g, 11 mmol) was reacted with (Z)-3-(3-chloro-2-fluoro-phenyl)-2-(4-chloro-2-fluoro-phenyl)-acrylonitrile (2.5 g, 8 mmol) prepared in Example 52a, AgF (0.7 g, 5.5 mmol), and triethylamine (2.9 g, 29 mmol) in dichloromethane (200 mL) at room temperature for 18 h to give rac-(2R,3S,4R,5S)-3-(3-Chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid tert-butyl ester as a white foam (3 g, 64%).
Example 52c Preparation of intermediate rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid trifluoroacetic acid
In a manner similar to the method described in Example 25a, rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid tert-butyl ester prepared in Example 52b (0.4 g, 0.8 mmol) was reacted with trifluoroacetic acid in dichloromethane at room temperature to give rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid trifluoroacetic acid as a white solid (0.5 g, 100%).
HRMS (ES+) m/z Calcd for C23H22Cl2F2N2O2+H [(M+H)+]: 467.1099, found: 467.1098.
Example 137 Preparation of rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid amide
In a manner similar to the method described in Examples 1e, rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid trifluoroacetic acid prepared in Example 52c (0.5 g, 0.86 mmol) was reacted with a dioxane solution (0.5 M) of ammonia (2 mL, 1 mmol), HATU (0.38 g, 1 mmol) and iPr2NEt (0.6 g, 4.6 mmol) in CH2Cl2 at room temperature for 20 h to give rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid amide as a white solid (0.3 g, 75%).
HRMS (ES+) m/z Calcd for C23H23Cl2F2N3O+H [(M+H)+]: 466.1259, found: 466.1259.
In a manner similar to the method described in Example 1b, 4-chloro-2-fluorophenylacetonitrile (5 g, 30 mmol) was reacted with 3-chloro-2-fluorobenzaldehyde (5 g, 32 mmol), methanolic solution (25 wt %) of sodium methoxide (21 mL, 92 mmol) in methanol (200 mL) at 45° C. for 5 h to give (Z)-3-(3-chloro-2-fluoro-phenyl)-2-(4-chloro-2-fluoro-phenyl)-acrylonitrile as a white powder (9 g, 97%).
Example 52b Preparation of intermediate rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid tert-butyl ester
In a manner similar to the method described in Example 1c, [3-methyl-but-(E)-ylideneamino]-acetic acid tert-butyl ester prepared in Example 1a (2.3 g, 11 mmol) was reacted with (Z)-3-(3-chloro-2-fluoro-phenyl)-2-(4-chloro-2-fluoro-phenyl)-acrylonitrile (2.5 g, 8 mmol) prepared in Example 52a, AgF (0.7 g, 5.5 mmol), and triethylamine (2.9 g, 29 mmol) in dichloromethane (200 mL) at room temperature for 18 h to give rac-(2R,3S,4R,5S)-3-(3-Chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid tert-butyl ester as a white foam (3 g, 64%).
Example 52c Preparation of intermediate rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid trifluoroacetic acid
In a manner similar to the method described in Example 25a, rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid tert-butyl ester prepared in Example 52b (0.4 g, 0.8 mmol) was reacted with trifluoroacetic acid in dichloromethane at room temperature to give rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid trifluoroacetic acid as a white solid (0.5 g, 100%).
HRMS (ES+) m/z Calcd for C23H22Cl2F2N2O2+H [(M+H)+]: 467.1099, found: 467.1098.
In a manner similar to the method described in Examples 1e, rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid trifluoroacetic acid prepared in Example 52c (0.5 g, 0.86 mmol) was reacted with a dioxane solution (0.5 M) of ammonia (2 mL, 1 mmol), HATU (0.38 g, 1 mmol) and iPr2NEt (0.6 g, 4.6 mmol) in CH2Cl2 at room temperature for 20 h to give rac-(2R,3S,4R,5S)-3-(3-chloro-2-fluoro-phenyl)-4-(4-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carboxylic acid amide as a white solid (0.3 g, 75%).
HRMS (ES+) m/z Calcd for C23H23Cl2F2N3O+H [(M+H)+]: 466.1259, found: 466.1259.

Example 447 Preparation of 4-{[(2R,3S,4R,5S)-4-(4-chloro-2-fluoro-phenyl)-3-(3-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carbonyl]-amino}-3-methoxy-benzoic acid methyl ester
In a 25 mL round-bottomed flask, (2R,3S,4R,5S)-3-(3-chloro-2-fluorophenyl)-4-(4-chloro-2-fluorophenyl)-4-cyano-5-neopentylpyrrolidine-2-carboxylic acid (250 mg, 535 μmol), was combined with CH2Cl2 (5 ml). DIPEA (277 mg, 374 μl, 2.14 mmol) and dipenylphospenic chloride (380 mg, 306 μl, 1.6 mmol) were added and the reaction was stirred at RT for 20 minutes. Methyl 4-amino-3-methoxybenzoate (100 mg, 552 μumol) was added and the reaction mixture was stirred at RT overnight.
The crude reaction mixture was concentrated in vacuum. The crude material was purified by flash chromatography (silica gel, 40 g, 5% to 25% EtOAc/Hexanes) to give the desired product as a white solid (275 mg, 81% yield).
Example 448 Preparation of 4-{[(2R,3S,4R,5S)-4-(4-Chloro-2-fluoro-phenyl)-3-(3-chloro-2-fluoro-phenyl)-4-cyano-5-(2,2-dimethyl-propyl)-pyrrolidine-2-carbonyl]-amino}-3-methoxy-benzoic acid
In a 25 mL round-bottomed flask, methyl 4-((2R,3S,4R,5S)-3-(3-chloro-2-fluorophenyl)-4-(4-chloro-2-fluorophenyl)-4-cyano-5-neopentylpyrrolidine-2-carboxamido)-3-methoxybenzoate (150 mg, 238 μmol, Eq: 1.00) was combined with CH2Cl2 (2 ml) to give a colorless solution. Aluminum bromide (Aldrich, 254 mg, 952 μmol, Eq: 4) and dimethyl sulfide (1.69 g, 2 mL, 27.2 mmol, Eq: 114) were added. The reaction mixture was stirred for overnight.
The reaction mixture was diluted with CH3CN (6 ml), EtOAc (10 ml) and water (10 ml), stirred and layers separated. The aqueous layer was extracted with EtOAc (2×10 mL). The organic layers were combined, washed with saturated NaCl (1×15 mL), dried over MgSO4 and concentrated in vacuum.
The crude material was dissolved in DMSO (4 ml) and was purified by preparative HPLC (70-100% ACETONITRILE/water). The fractions were combined, concentrated and freeze dried to give a white powder as desired product (75 mg, 51% yield). (ES+) m/z Calcd: [(M+H)+]: 616, found: 616.
Alternatively, the title compound could be prepared by the following method.
In a 500 mL round-bottomed flask, methyl 4-((2R,3S,4R,5S)-3-(3-chloro-2-fluorophenyl)-4-(4-chloro-2-fluorophenyl)-4-cyano-5-neopentylpyrrolidine-2-carboxamido)-3-methoxybenzoate (3.74 g, 5.93 mmol, Eq: 1.00) was combined with THF (140 ml) and MeOH (160 ml) at 50° C. to give a colorless solution. 1 N NaOH (23.7 ml, 23.7 mmol, Eq: 4) was added. The reaction mixture was stirred at 40° C. for 18 hrs.
The reaction mixture was concentrated to remove about ½ of the solvent, filtered to removed the insoluble, acidified with 1N HCl to PH=4-5 and the resulting solid was collected by filtration and was washed with water, small amount of MeOH and diethyl ether. It was then dried in vacuum oven (60° C.) overnight. Obtained was a white solid as the desired product (2.96 g, 80.5% yield). H1NMR and LC/MASS data were the same as that in the above procedure.
…………………………………………..
see
Bioorganic & Medicinal Chemistry (2014), 22(15), 4001-4009
| WO1996038131A1 * | May 30, 1996 | Dec 5, 1996 | James Matthew Butler | Method of producing a solid dispersion of a poorly water soluble drug |
| WO2010114928A2 * | Mar 31, 2010 | Oct 7, 2010 | F.Hoffmann-La Roche Ag | Compositions and uses thereof |
| WO2013139687A1 * | Mar 15, 2013 | Sep 26, 2013 | F. Hoffmann-La Roche Ag | Method for administration of an anti tumor agent |
| WO2013149981A1 * | Apr 2, 2013 | Oct 10, 2013 | F. Hoffmann-La Roche Ag | Pharmaceutical composition with improved bioavailability, safety and tolerability |
| CN102871950A * | Jul 15, 2011 | Jan 16, 2013 | 上海睿智化学研究有限公司 | 一种熊果酸固体分散体及其制备方法 |
| US20100152190 * | Feb 9, 2010 | Jun 17, 2010 | David Joseph Bartkovitz | Substituted Pyrrolidine-2-Carboxamides |
| US8354444 | Feb 9, 2010 | Jan 15, 2013 | Hoffmann-La Roche Inc. | Substituted pyrrolidine-2-carboxamides |
| US8709419 | Aug 10, 2011 | Apr 29, 2014 | Hoffmann-La Roche, Inc. | Combination therapy |
| US20130245089 * | Feb 5, 2013 | Sep 19, 2013 | Hoffmann-La Roche Inc. | Method for administration |
| WO2011098398A1 * | Feb 4, 2011 | Aug 18, 2011 | F. Hoffmann-La Roche Ag | Substituted pyrrolidine-2-carboxamides |
| WO2012007409A1 * | Jul 11, 2011 | Jan 19, 2012 | F. Hoffmann-La Roche Ag | N-substituted pyrrolidines |
| WO2013135648A1 | Mar 12, 2013 | Sep 19, 2013 | F. Hoffmann-La Roche Ag | Substituted pyrrolidine-2-carboxamides |
| WO2013139687A1 * | Mar 15, 2013 | Sep 26, 2013 | F. Hoffmann-La Roche Ag | Method for administration of an anti tumor agent |
| WO2013139724A1 | Mar 18, 2013 | Sep 26, 2013 | F. Hoffmann-La Roche Ag | Combination therapy (vemrufenib and a mdm2 inhibitor) for the treatment proliferative disorders |
| WO2013178570A1 | May 27, 2013 | Dec 5, 2013 | F. Hoffmann-La Roche Ag | Substituted pyrrolidine-2-carboxamides |
| WO2014114575A1 * | Jan 20, 2014 | Jul 31, 2014 | F. Hoffmann-La Roche Ag | Pharmaceutical composition with improved bioavailability |
REFERENCES
1 Discovery of RG7388, a Potent and Selective p53-MDM2 Inhibitor in Clinical Development. By Ding, Qingjie; Zhang, Zhuming; Liu, Jin-Jun; Jiang, Nan; Zhang, Jing; Ross, Tina M.; Chu, Xin-Jie; Bartkovitz, David; Podlaski, Frank; Janson, Cheryl; et al From Journal of Medicinal Chemistry (2013), 56(14), 5979-5983.
2. Pyrrolo[1,2-c]imidazolone derivatives as inhibitors of MDM2-p53 interactions and their preparation and use for the treatment of cancer. By Chu, Xin-Jie; Ding, Qingjie; Jiang, Nan; Liu, Jin-Jun; Ross, Tina Morgan; Zhang, Zhuming From U.S. Pat. Appl. Publ. (2012), US 20120065210 A1 20120315.
3. Pyrrolidine-2-carboxamide derivatives and their preparation and use as anticancer agents. By Chu, Xin-Jie; Ding, Qingjie; Jiang, Nan; Liu, Jin-Jun; Ross, Tina Morgan; Zhang, Zhuming. From U.S. Pat. Appl. Publ. (2012), US 20120010235 A1 20120112.
4. Preparation of substituted pyrrolidine-2-carboxamides as anticancer agents. By Bartkovitz, David Joseph; Chu, Xin-Jie; Ding, Qingjie; Jiang, Nan; Liu, Jin-Jun; Ross, Tina Morgan; Zhang, Jing; Zhang, Zhuming
From PCT Int. Appl. (2011), WO 2011098398 A1 20110818.
5. Preparation of substituted pyrrolidine-2-carboxamides as anticancer agents. By Bartkovitz, David Joseph; Chu, Xin-Jie; Ding, Qingjie; Jiang, Nan; Liu, Jin-Jun; Ross, Tina Morgan; Zhang, Jing; Zhang, Zhuming
From U.S. Pat. Appl. Publ. (2010), US 20100152190 A1 20100617.
6 B. Higgins, et al, Antitumor Activity of the MDM2 Antagonist RG7388, Mol Cancer Ther 2013;12(11 Suppl):B55
Discovery of RG7388, a potent and selective p53-MDM2 inhibitor in clinical development
J Med Chem 2013, 46(14): 5979


 STEREOCENTERS SHOWN
STEREOCENTERS SHOWN
There is an urgent medical need for novel antibacterial agents to treat hospital infections, specially those caused by multidrug-resistant Gram-positive pathogens. The need may also be fulfilled by either exploring antibacterial agents having new mechanism of action or expanding known classes of antibacterial drugs. The paper describes a new chemical entity, compound 21, derived from hitherto little known “floxacin”. The choice of the entity was made from a series of synthesized prodrugs and salts of the active chiral benzoquinolizine carboxylic acid, S-(−)-nadifloxacin. The chemistry, physicochemical characteristics, and essential bioprofile of 21 qualifies it for serious consideration as a novel drug entity against hospital infections of multi-drug-resistant Staphylococcus aureus, and its progress up to clinical phase I trials in humans is described.
quinolones has now grown to four generations, the first generation to nalidixic acid is represented as the representative of the second generation to PPA, only the Gram-negative bacteria effectively, the third generation is the development of these drugs the peak period, there has been a lot of drugs, and is a broad-spectrum antibiotic, which to norfloxacin, ciprofloxacin and other representatives. The fourth-generation quinolone antibiotics is in the third generation on the basis of a broad spectrum of antibacterial spectrum further expanded to make it available against mycoplasma and chlamydia infections.
[0003] R & D has been relatively popular domestic antibiotics, the most widely used on the market today is the third generation fluoroquinolones. Nadifloxacin developed by the Japanese company Otsuka, belongs to the third-generation quinolone antibacterial drugs, topical treatment of acne and folliculitis. 1993 for the first time in Japan (trade name: Acuatim), 2004 in the German market (trade name: Nadixa), 2005 in China listed (trade name: By Union, ointment).
[0004] nadifloxacin irritation due to its absorption and vascular problems, only made of topical formulations for in vitro Propionibacterium acnes (propionibacterium acnes) caused by acne. Wherein the S-(-) – that is the main role difloxacin isomer, the antibacterial activity of the R-isomer of 64 to 256 times that of racemic 2 times.
[0005] fine that gatifloxacin is S-(-) _ nadifloxacin salt on the basis of the system.Significantly improved solubility nadifloxacin well absorbed by the body, so it retains nadifloxacin broad spectrum antimicrobial, antibacterial activity, especially methicillin-sensitive Staphylococcus aureus and methicillin-resistant Staphylococcus aureus Effective characteristics (Antimicrobial Agents and Chemotherapy, 2004,3188 ~ 31920; J. Med. Chem. 2005 (48), 5232 ~ 5242). Pre-clinical tests prove that the product on the market anti-methicillin-resistant Staphylococcus aureus Antibiotic better compare the efficacy, including vancomycin, trovafloxacin, quinupristin + dalfopristin, linezolid amine.
[0006] fine molecular structure that gatifloxacin following formula:
[0007]
[0008] S-(-) _ nadifloxacin (C19H21FN2O4) with L-arginine salt, the further improve the play a major role in antibacterial s-(-) – nadifloxacin isomer content, and improved oral bioavailability, so that it can develop an oral or injectable preparations.
[0009] the literature (J. Med. Chem. 2005 (48), 5232 ~ 5242) discloses the synthesis of S_ (_) _ Nadifloxacin-L-arginine salt, S-(-) _ that fluoride gatifloxacin and L-arginine salt in the reaction solvent system, which solvent system is mainly methanol – water system, according to the paper reported in S-(-) – Nadifloxacin-L-arginine salt, yields were and related substances are not high enough.
Example 1
[0026] In equipped with oil bath, magnetic stirrer, thermometer, reflux condenser flask at 25 ° C was added (S) – (-) – nadifloxacin (100. 0g, 278mmol), dioxane ring (300ml), and the reaction solution was added dropwise to the L-arginine 4g, 278mmol) in distilled water (250ml) was added. Then heated to 50_60 ° C stirred 1.5 hours, and then adding activated carbon (3. Og) for 5 minutes, filtered hot, and then added dropwise at 55-60 ° C dioxane (700ml), and the natural cooling to 30 -35 ° C for 2 hours crystallization. The solid was collected by filtration and acetone (IOOml) wash. Dried at room temperature M hours. To give a white solid 137g, yield: 92%.
Example 1
Preparation of the single crystal of S-(- -9-fluoro-6,7-dihvdro-8-(4-hvdroxypiperidin-l-ylV5- methyl-l-oxo-lH,5H-benzo[“i,ι‘lquinolizine-2-carboxylic acid L-arginine salt terahvdrate.
S-(-)-9-Fluoro-6,7-dihydro-8-(4-hydroxypiperidin-l-yl)-5-methyl-l-oxo-lH,5H- benzo[i,j]quinolizine-2-carboxylic acid L-arginine salt (1.0 g) was dissolved in a mixture of acetone (40 ml) and water (10 ml) by heating the suspension at 70 °C for 15 minutes. The clear solution thus obtained was left for slow evaporation at room temperature in a beaker covered with a perforated aluminum foil. The crystal formation started after 2 days. Finally the single crystal was selected for X-ray crystal analysis from a cluster left after complete evaporation of the solvent. The ORTEP diagrams are described in Figures 1 and 2.
EXAMPLE 1
S-(-)-9-Fluoro-6,7-dihvdro-8-(4-hvdroxypiperidin-l-yl)-5-methyl-l-oxo-lH,5H-benzo Ti l quinolizine-2-carboxylic acid arginine salt Synthesis of SubstantiaUy CrystaUine product A solution of L-(+)-arginine (48.372 g, 0.278 mole) in distilled water (600 ml) was added dropwise over a period of 30 min to the stirred solution/suspension of finely powdered S-(-)-9-fluoro-6,7-dihydro-8-(4-hydroxypiperidin-l-yl)-5-methyl-l-oxo-lH,5H-benzo [ij] quinolizine-2-carboxylic acid (100 g, 0.278 mole) in acetone (1250 ml). The obtained clear solution was stirred for 30 min and concentrated on a water bath in vacuum (175 mbar) at 80°C. When product started solidifying, the concentration was carried out in vacuum (50 mbar) at 80°C up to dryness. Hexane (1 liter) was added, the reaction mixture was stirred for 4 hr, the solid thus separated was filtered and dried in vacuum (0.7 mbar) for 12 hrs at 70 °C. Yield 145 g (96.9%), m.p. 238-242 °C, and solubility 6 mg/ml (pH 9.5 buffer solution).
The substantially crystalline S-(-)-9-fluoro-6,7-dihydro-8-(4-hydroxypiperidin-l-yl)-5- methyl-l-oxo-lH,5H-benzo[i,j]quinolizine-2-carboxylic acid arginine salt prepared according to Example 1 possesses the following properties: a) Crystalline form, with a degree of crystallinity as determined by X-ray powder diffraction and as shown in Fig. 1. , b) A thermogram as determined by Differential scanning calorimetry and as shown in Fig. 3. c) Particle size measured as mean mass diameter (MMD) of 83.92 μm, as determined by laser diffraction technique. d) Density of 0.51 g/cm3 (untapped) and 0.7 g/cm3 (tapped). e) Hygroscopicity of 0% increase of weight upon storage for 14 days up to 22% relative atmospheric humidity as determined gravimetricaUy. f) A content of moisture water of 0.1 % by weight as determined by titration according to Karl Fischer. g) A content of acetone of 0.014 % by weight as determined by gas chromatography
Example 1
S-(-)-9-fluoro-6,7-dihydro-8-(4-hydroxypiperidin-l-yl)-5-methyI-l-oxo-lH,5H-benzo [ij] quinolizine-2-car boxy lie acid anhydrate
Method A
S-(-)-9-fluoro-6,7-dihydro-8-(4-hydroxypiperidin-l-yI)-5-methyl-l-oxo-lH,5H-benzo [ij] quinoIizine-2-carboxylic acid (3.0 g) obtained according to the process described in literature [K Hashimoto et al., Chem.Pharm.Bull.44, 642-5(1996)] was dissolved in acetonitrile (250 ml) at 85 °C. The resulting clear solution was filtered (to remove if any fibrous material is in suspension). The filtrate was concentrated to 125 ml and left at room temperature for crystallization. The crystals thus separated were filtered and dried in a drying cabinet at 40 °C for 2 hr in vacuum at 50 mm of Hg to obtain constant weight. Yield 2.6 g (86%).
Method B:
S-(-)-9-fluoro-6,7-dihydro-8-(4-hydroxypiperidin-l-yl)-5-methyI-l-oxo-lH,5H-benzo [ij] quinolizine-2-carboxyIic acid (2.0 g) obtained according to the process described in literature [K.Hashimoto etal., Chem.Pharm.Bull.44, 642-5(1996)] was dissolved in ethyl alcohol (95 %; 200 ml) at 80 °C. The obtained clear solution was filtered (to remove if any fibrous material is in suspension), concentrated to 100 ml and left for crystallization. The separated solid was Altered and dried in a drying cabinet at 40 °C for 3 hr in vacuum at 50 mm of Hg to obtain constant weight. Yield 1.7 g (85 %).
M.p.258-62 °C, moisture content 0 % (by Karl Fisher method) [CXJD 26 -299°, HPLC purity 99.8%
Example 8
S-(-)-9-fluoro-6,7-dihydro-8-(4-hydroxypiperidin-l-yl)-5-methyI-l-oxo-lH,5H-benzo [ij] quinolizine-2-carboxylic acid, L-arginine salt 0.75 hydrate
L-(+)-Arginine (0.958 g., 5.5 mmoles) was added in portions to a suspension solution of S- (-)-9-fluoro-6,7-dihydro-8-(4-hydroxypiperidin-l-yl)-5-methyl-l-oxo-lH,5H-benzo [ij] quinoIizine-2-carboxyIic acid 0.2 hydrate (2.0 g., 5.5 mmole) in methanol (400 ml). The obtained solution was concentrated in vacuum to give the desired product as a yellow solid, which was dried at 50 °C at 50 mm/Hg for 5 hours. Yield 3.0 g. (100%), m.p. 220- 223 °C (dec), m/z 535 (M+H), moisture content 2.3% (by Karl Fisher, required 2.46%), [CIJD 25 -144 ° (1% methanol c=l), solubility 93 mg/ml.
……………………………..
https://www.jstage.jst.go.jp/article/cpb1958/44/4/44_4_642/_article
(S)-(-)-Nadifloxacin [(S)-(-)-9-fluoro-6, 7-dihydro-8-(4-hydroxy-1-piperidyl)-5-methyl-1-oxo-1H, 5H-benzo[i, j]quinolizine-2-carboxylic acid, (S)-(-)-OPC-7251], an antibacterial agent, was synthesized from (S)-(-)-5, 6-difluoro-2-methyl-1, 2, 3, 4-tetrahydroquinoline (DFTQ), which was prepared by the optical resolution of recemic DFTQ with 2, 3-di-O-benzoxyl-L-tartaric acid. Racemization of the undesired enantiomer [(R)-(+)-DFTQ] was studied in the presence of various acids and the best result was obtained in the case of methanesulfonic acid. The absolute configuration of (-)-nadifloxacin was determined as S by X-ray crystallographic analysis.
https://www.jstage.jst.go.jp/article/cpb1958/44/4/44_4_642/_pdf ………..FREE PDF

 A Chiral Benzoquinolizine-2-carboxylic acid Arginine Salt Active against Vancomycin Intermediate Staphylococcus aureus (VISA). Abstract of Papers, 43rd Interscience Conference on Antimicrobial Agents and Chemotherapy, Chicago, September 2003;American Society for Microbiology: Washington, DC, 2003; Poster F-430
A Chiral Benzoquinolizine-2-carboxylic acid Arginine Salt Active against Vancomycin Intermediate Staphylococcus aureus (VISA). Abstract of Papers, 43rd Interscience Conference on Antimicrobial Agents and Chemotherapy, Chicago, September 2003;American Society for Microbiology: Washington, DC, 2003; Poster F-430Some quinolones introduced for clinical use.


Ambroxol is a secretolytic agent used in the treatment of respiratory diseases associated with viscid or excessive mucus. It is the active ingredient of Mucosolvan, Mucobrox, Mucol, Lasolvan, Mucoangin, Surbronc, Ambolar, and Lysopain. The substance is a mucoactive drug with several properties including secretolytic and secretomotoric actions that restore the physiological clearance mechanisms of the respiratory tract, which play an important role in the body’s natural defence mechanisms. It stimulates synthesisand release of surfactant by type II pneumocytes. Surfactant acts as an anti-glue factor by reducing the adhesion of mucus to thebronchial wall, in improving its transport and in providing protection against infection and irritating agents.[1][2] Ambroxol is often administered as an active ingredient in cough syrup.
Ambroxol is indicated as “secretolytic therapy in bronchopulmonary diseases associated with abnormal mucus secretion and impaired mucus transport. It promotes mucus clearance, facilitates expectoration and eases productive cough, allowing patients to breathe freely and deeply”.[3]

Ambroxolhydrochloridetablets in Japan
There are many different formulations developed since the first marketing authorisation in 1978. Ambroxol is available as syrup, tablets, pastilles, dry powder sachets, inhalation solution, drops and ampules as well aseffervescent tablets.
Ambroxol also provides pain relief in acute sore throat. Pain in sore throat is the hallmark of acutepharyngitis.[4] Sore throat is usually caused by a viral infection. The infection is self limited and the patient recovers normally after a few days. What is most bothering for the patient is the continuous pain in the throat maximized when the patient is swallowing. The main goal of treatment is thus to reduce pain. The main property of Ambroxol for treating sore throat is the local anaesthetic effect, described first in the late 1970s,[5][6] but explained and confirmed in more recent work.
Ambroxol is a potent inhibitor of the neuronal Na+ channels.[7] This property led to the development of alozenge containing 20 mg of ambroxol. Many state-of-the-art clinical studies[4] have demonstrated the efficacy of Ambroxol in relieving pain in acute sore throat, with a rapid onset of action, with its effect lasting at least three hours. Ambroxol is also anti-inflammatory, reducing redness in a sore throat.
Ambroxol has recently been shown to increase activity of the lysosomal enzyme glucocerebrosidase. Because of this it may be a useful therapeutic agent for both Gaucher disease and Parkinson’s disease.
Ambroxol is a secretolytic agent used in the treatment of respiratory diseases associated with viscid or excessive mucus. It is the active ingredient of Mucosolvan, Lasolvan or Mucoangin. The substance is a mucoactive drug with several properties including secretolytic and secretomotoric actions that restore the physiological clearance mechanisms of the respiratory tract which play an important role in the body’s natural defence mechanisms. It stimulates synthesis and release of surfactant by type II pneumocytes. Surfactants acts as an anti-glue factor by reducing the adhesion of mucus to the bronchial wall, in improving its transport and in providing protection against infection and irritating agents.
| Salt | ATC | Formula | MM | CAS |
|---|---|---|---|---|
| – | R02AD05 R05CB06 R07AA03 |
C 13 H 18 Br 2 N 2 O | 378.11 g / mol | 18683-91-5 |
 |
|
| Systematic (IUPAC) name | |
|---|---|
| trans-4-(2-Amino-3,5-dibrombenzylamino)-cyclohexanol | |
| Clinical data | |
| AHFS/Drugs.com | International Drug Names |
| Identifiers | |
| ATC code | R05CB06 |
| PubChem | CID 2132 |
| ChemSpider | 10276826 |
| UNII | 200168S0CL |
| KEGG | D07442 |
| ChEMBL | CHEMBL153479 |
| Chemical data | |
| Formula | C13H18Br2N2O |
| Mol. mass | 378.10 |
| Синтез a) |
|---|
  |

Ambroxol synthesis.[9]
| melting point | 233-234.5 | Kack, J., Koss, F.W., Schraven, E. and Beisenherz, G.; US. Patent 3,536,713; October 27, 1970; assigned to Boehringer lngelheim G.m.b.H. |
Kack, J., Koss, F.W., Schraven, E. and Beisenherz, G.; US. Patent 3,536,713; October 27,
1970; assigned to Boehringer lngelheim G.m.b.H.
| Page | Trade name | Manufacturer |
|---|---|---|
| Germany | Ambrobeta | betapharm |
| AmbroGEKSAL | Hexal | |
| Mucosal | Boehringer Ingelheim | |
| various generic drugs | ||
| France | Muksol | Leurquin Milan |
| Surbronk | Boehringer Ingelheim | |
| Italy | Ambrotus | Epifarma |
| ATUS | Metapharma | |
| Mukoarikodil | Menarini | |
| Mucosal | Boehringer Ingelheim, 1982 | |
| Viskomucil | Institute of Organic Chem. | |
| Japan | Mucosal | Teijin |
| Ukraine | AMBROKSOL | Ltd. “Pilot Plant” HNTSLS “m. Kharkiv, Ukraine |
| Ambroxol hydrochloride | CJC BHFZ, m. Kyiv, Ukraine | |
| AMBROBENE | ratiopharm GmbH, Germany | |
| LAZOLVAN® | Boehringer Ingelheim International GmbH, Germany | |
| various generic drugs | ||

The Japanese government said this week that it is prepared to make an influenza drug that is not approved for the treatment of Ebola available to West African countries hard-hit by the deadly virus.
http://cen.acs.org/articles/92/i35/Japan-Proposes-Influenza-Drug-Treat.html
Pharmaceuticals: Country says Fujifilm’s favipiravir is available.
KEY
Fujifilm’s, favipiravir, EBOLA

Telmapitant
TELMAPITANT; Telmapitant (USAN); Telmapitant [USAN]; 552292-58-7; HJ5FE4153B; D10391
(5R,8S)-8-[[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]methyl]-8-phenyl-1,3,9-triazaspiro[4.5]decane-2,4-dione
1,3,7-Triazaspiro[4.5]decane-2,4-dione, 8-[[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]methyl]-8-phenyl-, (5R,8S)-
(5R,8S)-8-(((1R)-1-(3,5-Bis(Trifluoromethyl)phenyl)ethoxy)methyl)-8-phenyl-1,3,7- triazaspiro(4.5)decane-2,4-dione
1,3,7-Triazaspiro(4.5)decane-2,4-dione,
8-(((1R)-1-(3,5-bis(trifluoromethyl)phenyl)ethoxy)methyl)-8-phenyl-, (5R,8S)-
Molecular Formula: C24H23F6N3O3
Molecular Weight: 515.448139
cas 552292-58-7
Merck & Co. (innovator)
Treatment of Nausea and Vomiting,
SYNTHESIS
……………………………………….
US7902366
http://www.google.com/patents/US7902366
Example 43a Example 43b
Step 1:
To a suspension of lactol Compound 3 (60 g, 93.0 mmol, 1 equiv.) and Wittig Reagent (93.5 g, 200.0 mmol, 2.15 equiv.) in toluene (800 ml) stirred at −78° C. under N2, a solution of KHMDS (0.5M in toluene, 558 ml, 280.0 mmol, 3 equiv.) was added dropwise at −78° C. The cooling bath was removed and the yellow mixture was warmed to RT to form a red solution. The mixture was allowed to stir at 23° C. for further 1 h before being quenched with saturated NH4Cl solution. EtOAc was added and layers were separated. The separated aqueous layer was extracted with EtOAc (2×500 ml). The combined organic layers were dried (MgSO4) and filtered. Removal of solvents in vacuum followed by Biotage column chromatography [5% EtOAc-hexane to 10% EtOAc-hexane] gave alkene Compound 42 as white solid (40.5 g, 68%), Electrospray MS [M+1]+ 638.1. Continuous elution gave an impure cyclized product Compound 43.
Step 2:
A suspension of alkene Compound 42 (40.5 g, 64 mmol, 1 equiv.) and PtO2 (1.44 g, 6.4 mmol, 0.1 equiv.) in EtOH (400 ml) were stirred under a H2 balloon at 23° C. for 24 h. Another batch of PtO2 (1.44 g, 6.4 mmol, 0.1 equiv) was added and the mixture was stirred for another 24 h at 23° C. The catalyst was filtered via a pad of Celite. This solution of alkane Compound 44 was used in the next step without further purification.
Step 3:
p-TsOH.H2O (2.42 g, 13.0 mmol) was added to the ethanolic solution of alkane Compound 44 from above and the solution was heated to reflux for 4 h. The solution was cooled to RT and neutralized with Et3N. Solvents were removed in vacuum and EtOAc was added. Saturated NaHCO3 solution was added and layers were separated. The separated aqueous layer was extracted with EtOAc (300 ml×2). The combined organic layers were dried (MgSO4) and filtered. Removal of solvents in vacuum followed by Biotage column chromatography [10% ether-hexane] gave enamide Compound 45 (first batch) as yellow oil. Some intermediate and starting material were recovered as yellow oil by continuous elution with [50% EtOAc-hexane]. The yellow oil was dissolved in toluene and 10 mol % p-TsOH was added. The mixture was heated to reflux for 2 h and cooled to RT. Work up was as above and the combined enamide Compound 45 (25 g, 70%), Electrospray MS [M+1]+ 564.1, was obtained as yellow oil.
Step 4:
BH3.Me2S (13.6 ml, 133 mmo, 3.02 equiv) was added to a solution of enamide Compound 45 (25 g, 44.0 mmol,1 equiv.) in THF at 23° C. under N2. The mixture was stirred at 23° C. for 18 h and then cooled over an ice-water bath. A solution of NaOH (500 ml, 2N) was added slowly followed by a solution of H202 (500 ml, 30% aqueous). The mixture was allowed to stir from 0° C. to 23° C. for 18 h. Layers were separated and the separated aqueous layer was extracted with Et2O (500 ml×2). The combined organic layers were dried (MgSO4) and filtered. Removal of solvents in vacuum followed by Biotage column chromatography [hexane-EtOAc, 3:1 (v/v)] gave alcohol Compound 46 as colorless oil (19 g, 74%), Electrospray MS [M+1]+ 582.1.
Step 5:
Oxalyl chloride (5.7 ml, 65.3 mmol, 2 equiv.) was added to a solution of DMSO (9.3 ml, 131.0 mmol, 4 equiv.) in CH2Cl2 (300 ml) at −78° C. under N2. The mixture was stirred at −78° C. for 15 min before a solution of alcohol Compound 46 (19 g, 32.7 mmol. 1 equiv.) in CH2Cl2 (50 ml) was added. The mixture was stirred at −78° C. for a further 1 h and Et3N (32 ml, 228.9 mmol, 7 equiv.) was added. The cooling bath was removed and the mixture was warmed to RT before it was quenched with saturated NaHCO3 solution. Layers were separated and the aqueous was extracted with CH2Cl2 (300 ml×2). The combined organic layers were dried (MgSO4) and filtered. Removal of solvents in vacuum followed by Biotage column chromatography [hexane-ether, 4:1 (v/v)] gave ketone Compound 47 as colorless oil (15 g, 80%), Electrospray MS [M+1]+ 580.1.
Step 6:
EtOH (150 ml) was added to Cbz-ketone Compound 47 (15 g, 25.88 mmol, 1 equiv.), followed by NH4(CO3)2 (9.95 g, 103.5 mmol, 4 equiv.) and a solution of KCN (3.4 g, 51.77 mmol, 2 equiv.). The resulting mixture was heated at 58° C. under N2 for 72 h. TLC (1:1 EtOAc:hexane) revealed complete consumption of the starting material. The reaction mixture was cooled to RT and poured into sat. aq. NaHCO3 (200 ml) and extracted with EtOAc (3×200 ml). The combined organic layers were dried over MgSO4 and concentrated in vacuo to afford crude Cbz-hydantoin Compound 48 (16.5 g, 98%), Electrospray MS [M+1]+650.1. The crude material was used in the next reaction without further purification.
Step 7:
The crude Cbz-hydantoin Compound 48 (16.5 g, 25.4 mmol, 1 equiv.) was dissolved in MeOH (220 ml) and 20% Pd(OH)2—C (3.6 g) was added. The reaction mixture was shaken in a parr shaker under H2 atmosphere at 40 psi for 18 h. TLC (1:1 EtOAc:hexane) revealed complete consumption of the starting material. The reaction mixture was filtered through a pad of celite and the celite was washed with MeOH. The resulting solution was concentrated in vacuo. The crude product was purified by column chromatography on a Biotage (3:2, EtOAc:hex). Two major spots were collected. The less-polar spot corresponds to the isomer Example 43a (3 g, overall 20% over two steps), Electrospray MS [M+1]+ 516.1. The more polar spot corresponds to the isomer Example 43b (4.5 g, overall 30% over two steps), Electrospray MS [M+1]+ 516.1.
………………………………..
http://www.google.com/patents/WO2003051840A1?cl=en
Example 43a Example 43b
Step 1 :
Compound 3
To a suspension of lactol Compound 3 (60g, 93.0mmol, lequiv.) and Wittig Reagent (93. δg, 200.0mmol, 2.1 δequiv.) in toluene (800ml) stirred at -78°C under δ N2, a solution of KHMDS (O.δM in toluene, δδδml, 280.0mmol, 3equiv.) was added dropwise at -78°C. The cooling bath was removed and the yellow mixture was warmed to RT to form a red solution. The mixture was allowed to stir at 23°C for further 1 h before being quenched with saturated NH CI solution. EtOAc was added and layers were separated. The separated aqueous layer was extracted with EtOAc 0 (2 x δOOml). The combined organic layers were dried (MgSO ) and filtered.
Removal of solvents in vacuum followed by Biotage column chromatography [δ% EtOAc-hexane to 10% EtOAc-hexane] gave alkene Compound 42 as white solid (40. δg, 68%), Electrospray MS [M+1]+ 638.1. Continuous elution gave an impure cyclized product Compound 43. δ Step 2:
Compound 42
A suspension of alkene Compound 42 (40. δg, 64mmol, lequiv.) and PtO2 (1.44g, 6.4mmol, 0.1 equiv.) in EtOH (400ml) were stirred under a H2 balloon at 23°C for 24 h. Another batch of PtO2 (1.44g, 6.4mmol, 0.1 equiv) was added and the 0 mixture was stirred for another 24 h at 23°C. The catalyst was filtered via a pad of Celite. This solution of alkane Compound 44 was used in the next step without further purification. Step 3:
Compound 44
p-TsOH.H2O (2.42g, 13.0mmol) was added to the ethanolic solution of alkane
Compound 44 from above and the solution was heated to reflux for 4 h. The solution was cooled to RT and neutralized with Et3N. Solvents were removed in vacuum and EtOAc was added. Saturated NaHCO3 solution was added and layers
5 were separated. The separated aqueous layer was extracted with EtOAc (300ml x
2). The combined organic layers were dried (MgSO4) and filtered. Removal of solvents in vacuum followed by Biotage column chromatography [10% ether- hexane] gave enamide Compound 45 (first batch) as yellow oil. Some intermediate and starting material were recovered as yellow oil by continuous elution with 0 [50%EtOAc-hexane]. The yellow oil was dissolved in toluene and 10mol% p-TsOH was added. The mixture was heated to reflux for 2 h and cooled to RT. Work up was as above and the combined enamide Compound 45 (2δg, 70%), Electrospray
MS [M+1]+ 664.1 , was obtained as yellow oil.
Step 4:
BH3.Me2S (13.6ml, 133mmo, 3.02 equiv) was added to a solution of enamide Compound 45T25g, 44.0mmol, lequiv.) in THF at 23°C under N2. The mixture was stirred at 23°C for 18 h and then cooled over an ice-water bath. A solution of NaOH (600ml, 2N) was added slowly followed by a solution of H O2 (600ml, 30% 0 aqueous). The mixture was allowed to stir from 0°C to 23°C for 18 h. Layers were separated and the separated aqueous layer was extracted with Et.20 (600ml x 2). The combined organic layers were dried (MgSO4) and filtered. Removal of solvents in vacuum followed by Biotage column chromatography [hexane-EtOAc, 3:1 (v/v)] gave alcohol Compound 46 as colorless oil (19g, 74%), Electrospray MS [M+1]+ δ 582.1. Step 5:
Compound 46
Oxalyl chloride (δ.7ml, 6δ.3mmol, 2equiv.) was added to a solution of DMSO (9.3ml, 131.0mmol, 4equiv.) in CH2CI2 (300ml) at -78°C under N2. The mixture was 0 stirred at -78°C for 1 δ min before a solution of alcohol Compound 46 (19g, 32.7mmol. lequiv.) in CH2CI2 (50ml) was added. The mixture was stirred at -78°C for a further 1 h and Et3N (32ml, 228.9mmol, 7equiv.) was added. The cooling bath was removed and the mixture was warmed to RT before it was quenched with saturated NaHCO3 solution. Layers were separated and the aqueous was extracted with CH2CI2 (300ml x 2). The combined organic layers were dried (MgSO4) and filtered. Removal of solvents in vacuum followed by Biotage column chromatography [hexane-ether, 4:1 (v/v)] gave ketone Compound 47 as colorless oil (1δg, 80%), Electrospray MS [M+1]+ 680.1.
EtOH (150ml) was added to Cbz-ketone Compound 47 (15g, 2δ.88mmol, lequiv.), followed by NH (CO )2 (9.9δg, 103.5mmol, 4equiv.) and a solution of KCN (3.4g, 61.77mmoI, 2equiv.). The resulting mixture was heated at 68°C under N2 for 72 h. TLC (1 :1 EtOAc:hexane) revealed complete consumption of the starting
1δ material. The reaction mixture was cooled to RT and poured into sat. aq. NaHCO3 (200 ml) and extracted with EtOAc (3 x 200ml). The combined organic layers were dried over MgSO4 and concentrated in vacuo to afford crude Cbz-hydantoin Compound 48 (16.δg, 98%), Electrospray MS [M+1]+ 650.1. The crude material was used in the next reaction without further purification.
20 Step 7:
The crude Cbz-hydantoin Compound 48 (16.5g, 2δ.4mmol, lequiv.) was dissolved in MeOH (220ml) and 20% Pd(OH)2-C (3.6g) was added. The reaction mixture was shaken in a parr shaker under H2 atmosphere at 40 psi for 18 h. TLC (1 :1 EtOAc:hexane) revealed complete consumption of the starting material. The
26 reaction mixture was filtered through a pad of celite and the celite was washed with MeOH. The resulting solution was concentrated in vacuo. The crude product was purified by column chromatography on a Biotage (3:2, EtOAc:hex). Two major spots were collected. The less-polar spot corresponds to the isomer Example 43a (3 g, overall 20% over two steps), Electrospray MS [M+1]+ 616.1. The more polar spot
30 corresponds to the isomer Example 43b (4.6 g, overall 30% over two steps), Electrospray MS [M+1]+ 616.1.
|
4-29-2011
|
NK1 ANTAGONISTS
|
|
|
3-9-2011
|
NK1 antagonists
|
| English translation of Knabe, J., et al., “Racemates and Enantiomers of . . . ,” Pharmazie 52(12):912-919 (1997). | ||
| 2 | English translation of Schult, Karl E., et al., “Hydantoin-Derivate as Potential . . . ,” Eur. J. Med. Chem.-Chimica Therapeutics 13(1):25-31 (1978). | |
| 3 | English translation of Schult, Karl E., et al., “Hydantoin-Derivate as Potential . . . ,” Eur. J. Med. Chem.—Chimica Therapeutics 13(1):25-31 (1978). | |
| 4 | Knabe, J., et al., “Racemates and Enantiomers of Basic Substituted 5-Phenylhydantoins . . . ,” Pharmazie 52(12): 912-919 (1997). | |
| 5 | Oh, Chang-Hyun et al., “Synthesis of New Hydantoin-3-Acetic Acid Derivatives . . . ,” Bull. Korean Chem. Soc. 9(4):231-235 (1988). | |
| 6 | Shulte, Karl E., et al., “Hydantoin-Derivate als . . . ,” Eur. J. Med. Chem.-Chimica Therapeutica 13(1):25-31 (1978). | |
| 7 | Shulte, Karl E., et al., “Hydantoin-Derivate als . . . ,” Eur. J. Med. Chem.—Chimica Therapeutica 13(1):25-31 (1978). | |
| 8 | Wu, X. et al., “Generation of Cyclopenta [c] piperidines and Pyrrolo [3,4-c]piperidines- . . . ,” Tetrahedron 56(34): 6279-6290 (2000). | |
| 9 | * | Xiujuan Wu et al 2000. , Stereoselective transformation of 2H-1,4-Oxazin-2-ones into 2,(2),5,5-tri- and tetrasubstituted Analogues. . . |
| US6436928 * | Dec 14, 2000 | Aug 20, 2002 | Schering Corporation | Selective neurokinin antagonists |
| US6635639 * | Feb 13, 2002 | Oct 21, 2003 | Nps Allelix Corp. | Use of N-alkylamino-heterocylic compounds for the treatment of migraine |
| US7041682 * | Jul 2, 2003 | May 9, 2006 | Schering Corporation | Antiemetics, antidepressants, anxiolytic agents, antitussive agents |
| US7122677 * | Nov 12, 2002 | Oct 17, 2006 | Scherig Corporation | NK1 antagonists |
| US20060094720 * | Dec 15, 2005 | May 4, 2006 | Neng-Yang Shih | NK1 antagonists |
| US20060223804 * | Jun 30, 2005 | Oct 5, 2006 | Schering Corporation | NK1 antagonists |
| EP0790248A1 | Jan 20, 1997 | Aug 20, 1997 | Pfizer Limited | 3-Aza-piperidone- (tetrahydropyrimidin-2-one) and 3-oxa-piperidone (1,3 oxazin-2-one) derivatives, their preparation and their use as tachykinin/neurokinin antagonists |
key
Telmapitant, Merck, Tachykinin NK1 Antagonists

Aldoxorubicin, DOXO-EMCH
N’-[1-[4(S)-(3-Amino-2,3,6-trideoxy-alpha-L-lyxo-hexopyranosyloxy)-2(S),5,12-trihydroxy-7-methoxy-6,11-dioxo-1,2,3,4,6,11-hexahydronaphthacen-2-yl]-2-hydroxyethylidene]-6-(2,5-dioxo-2,5-dihydro-1H-pyrrol-1-yl)hexanohydrazide
1H-Pyrrole-1-hexanoic acid, 2,5-dihydro-2,5-dioxo-, (2E)-2-[1-[(2S,4S)-4-[(3-amino-2,3,6-trideoxy-α-L-lyxo– hexopyranosyl)oxy]-1,2,3,4,6,11-hexahydro-2,5,12- trihydroxy-7-methoxy-6,11-dioxo-2-naphthacenyl]-2- hydroxyethylidene]hydrazide
CytRx is pouring money into R&D of cancer-fighting drugs see article
Los Angeles Times
s most promising cancer-fighting drug, aldoxorubicin, is “sort of like a guided … Phase 3 clinical trial of a second-line treatment for soft-tissue sarcoma.

Aldoxorubicin
http://www.ama-assn.org/resources/doc/usan/aldoxorubicin.pdf
 in phase 3 Cytrx Corporation
in phase 3 Cytrx Corporation
(E)-N’-(1-((2S,4S)-4-(((2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)-2,5,12-trihydroxy-7-methoxy-6,11-dioxo-1,2,3,4,6,11-hexahydrotetracen-2-yl)-2-hydroxyethylidene)-6-(2,5-dioxo-2,5-dihydro-1H-pyrrol-1-yl)hexanehydrazide hydrochloride
1H-Pyrrole-1-hexanoic acid, 2,5-dihydro-2,5-dioxo-, (2E)-2-[1-[(2S,4S)-4-[(3-amino-
2,3,6-trideoxy-α-L-lyxo-hexopyranosyl)oxy]-1,2,3,4,6,11-hexahydro-2,5,12-trihydroxy-
7-methoxy-6,11-dioxo-2-naphthacenyl]-2-hydroxyethylidene]hydrazide
N’-[(1E)-1-{(2S,4S)-4-[(3-amino-2,3,6-trideoxy-α-L-lyxo-hexopyranosyl)oxy]-2,5,12-
trihydroxy-7-methoxy-6,11-dioxo-1,2,3,4,6,11-hexahydrotetracen-2-yl}-2-
hydroxyethylidene]-6-(2,5-dioxo-2,5-dihydro-1H-pyrrol-1-yl)hexanohydrazide
MOLECULAR FORMULA C37H42N4O13
MOLECULAR WEIGHT 750.7
SPONSOR CytRx Corp.
CODE DESIGNATION
CAS REGISTRY NUMBER 1361644-26-9
CAS: 151038-96-9 (INNO-206); 480998-12-7 (INNO-206 HCl salt), 1361644-26-9
| QC data: | |
| Safety Data Sheet (MSDS): |

hydrochloride
CAS: 151038-96-9
Chemical Formula: C37H42N4O13
Exact Mass: 750.27484
Molecular Weight: 750.75
| Certificate of Analysis: | |
| QC data: | |
| Safety Data Sheet (MSDS): |

| In vitro protocol: | Clin Cancer Res. 2012 Jul 15;18(14):3856-67 |
| In vivo protocol: | Clin Cancer Res. 2012 Jul 15;18(14):3856-67.Invest New Drugs. 2010 Feb;28(1):14-9.Invest New Drugs. 2012 Aug;30(4):1743-9.Int J Cancer. 2007 Feb 15;120(4):927-34. |
| Clinical study: | Expert Opin Investig Drugs. 2007 Jun;16(6):855-66. |

Aldoxorubicin (INNO-206): Aldoxorubicin, also known as INNO-206, is the 6-maleimidocaproyl hydrazone derivative prodrug of the anthracycline antibiotic doxorubicin (DOXO-EMCH) with antineoplastic activity. Following intravenous administration, doxorubicin prodrug INNO-206 binds selectively to the cysteine-34 position of albumin via its maleimide moiety. Doxorubicin is released from the albumin carrier after cleavage of the acid-sensitive hydrazone linker within the acidic environment of tumors and, once located intracellularly, intercalates DNA, inhibits DNA synthesis, and induces apoptosis. Albumin tends to accumulate in solid tumors as a result of high metabolic turnover, rapid angiogenesis, hyervasculature, and impaired lymphatic drainage. Because of passive accumulation within tumors, this agent may improve the therapeutic effects of doxorubicin while minimizing systemic toxicity.
“Aldoxorubicin has demonstrated effectiveness against a range of tumors in both human and animal studies, thus we are optimistic in regard to a potential treatment for Kaposi’s sarcoma. The current standard-of-care for severe dermatological and systemic KS is liposomal doxorubicin (Doxil®). However, many patients exhibit minimal to no clinical response to this agent, and that drug has significant toxicity and manufacturing issues,” said CytRx President and CEO Steven A. Kriegsman. “In addition to obtaining valuable information related to Kaposi’s sarcoma, this trial represents another opportunity to validate the value and viability of our linker technology platform.” The company expects to announce Phase-2 study results in the second quarter of 2015.
Kaposi’s sarcoma is an orphan indication, meaning that only a small portion of the population has been diagnosed with the disease (fewer than 200,000 individuals in the country), and in turn, little research and drug development is being conducted to treat and cure it. The FDA’s Orphan Drug Act may grant orphan drug designation to a drug such as aldoxorubicin that treats a rare disease like Kaposi’s sarcoma, offering market exclusivity for seven years, fast-track status in some cases, tax credits, and grant monies to accelerate research
The widely used chemotherapeutic agent doxorubicin is delivered systemically and is highly toxic, which limits its dose to a level below its maximum therapeutic benefit. Doxorubicin also is associated with many side effects, especially the potential for damage to heart muscle at cumulative doses greater than 450 mg/m2. Aldoxorubicin combines doxorubicin with a novel single-molecule linker that binds directly and specifically to circulating albumin, the most plentiful protein in the bloodstream. Protein-hungry tumors concentrate albumin, thus increasing the delivery of the linker molecule with the attached doxorubicin to tumor sites. In the acidic environment of the tumor, but not the neutral environment of healthy tissues, doxorubicin is released. This allows for greater doses (3 1/2 to 4 times) of doxorubicin to be administered while reducing its toxic side effects. In studies thus far there has been no evidence of clinically significant effects of aldoxorubicin on heart muscle, even at cumulative doses of drug well in excess of 2,000 mg/m2.
INNO-206 is an anthracycline in early clinical trials at CytRx Oncology for the treatment of breast cancer, HIV-related Kaposi’s sarcoma, glioblastoma multiforme, stomach cancer and pancreatic cancer. In 2014, a pivotal global phase 3 clinical trial was initiated as second-line treatment in patients with metastatic, locally advanced or unresectable soft tissue sarcomas. The drug candidate was originally developed at Bristol-Myers Squibb, and was subsequently licensed to KTB Tumorforschungs. In August 2006, Innovive Pharmaceuticals (acquired by CytRx in 2008) licensed the patent rights from KTB for the worldwide development and commercialization of the drug candidate. No recent development has been reported for research that had been ongoing for the treatment of small cell lung cancer (SCLC).
INNO-206 is a doxorubicin prodrug. Specifically, it is the 6-maleimidocaproyl hydrazone of doxorubicin. After administration, the drug candidate rapidly binds endogenous circulating albumin through the acid sensitive EMCH linker. Circulating albumin preferentially accumulates in tumors, bypassing uptake by other non-specific sites including the heart, bone marrow and the gastrointestinal tract. Once inside the acidic environment of the tumor cell, the EMCH linker is cleaved and free doxorubicin is released at the tumor site. Like other anthracyclines, doxorubicin inhibits DNA and RNA synthesis by intercalating between base pairs of the DNA/RNA strand, thus preventing the replication of rapidly-growing cancer cells. It also creates iron-mediated free oxygen radicals that damage the DNA and cell membranes. In 2011, orphan drug designation was assigned in the U.S. for the treatment of pancreatic cancer and for the treatment of soft tissue sarcoma.
CytRx Corporation (NASDAQ:CYTR) has announced it has initiated a pivotal global Phase 3 clinical trial to evaluate the efficacy and safety of aldoxorubicin as a second-line treatment for patients with soft tissue sarcoma (STS) under a Special Protocol Assessment with the FDA. Aldoxorubicin combines the chemotherapeutic agent doxorubicin with a novel linker-molecule that binds specifically to albumin in the blood to allow for delivery of higher amounts of doxorubicin (3.5 to 4 times) without several of the major treatment-limiting toxicities seen with administration of doxorubicin alone.
According to a news from Medicalnewstoday.com; CytRx holds the exclusive worldwide rights to INNO-206. The Company has previously announced plans to initiate Phase 2 proof-of-concept clinical trials in patients with pancreatic cancer, gastric cancer and soft tissue sarcomas, upon the completion of optimizing the formulation of INNO-206. Based on the multiple myeloma interim results, the Company is exploring the possibility of rapidly including multiple myeloma in its INNO-206 clinical development plans.
According to CytRx’s website, In preclinical models, INNO-206 was superior to doxorubicin with regard to ability to increase dosing, antitumor efficacy and safety. A Phase I study of INNO-206 that demonstrated safety and objective clinical responses in a variety of tumor types was completed in the beginning of 2006 and presented at the March 2006 Krebskongress meeting in Berlin. In this study, doses were administered at up to 4 times the standard dosing of doxorubicin without an increase in observed side effects over historically seen levels. Objective clinical responses were seen in patients with sarcoma, breast, and lung cancers.
INNO-206 – Mechanism of action:
According to CytRx’s website, the proposed mechanism of action is as the follow steps: (1) after administration, INNO-206 rapidly binds endogenous circulating albumin through the EMCH linker. (2) circulating albumin preferentially accumulates in tumors, bypassing uptake by other non-specific sites including heart, bone marrow and gastrointestinal tract; (3) once albumin-bound INNO-206 reaches the tumor, the acidic environment of the tumor causes cleavage of the acid sensitive linker; (4) free doxorubicin is released at the site of the tumor.
INNO-206 – status of clinical trials:
CytRx has announced that, in December 2011, CytRx initiated its international Phase 2b clinical trial to evaluate the preliminary efficacy and safety of INNO-206 as a first-line therapy in patients with soft tissue sarcoma who are ineligible for surgery. The Phase 2b clinical trial will provide the first direct clinical trial comparison of INNO-206 with native doxorubicin, which is dose-limited due to toxicity, as a first-line therapy. (source:http://cytrx.com/inno_206, accessed date: 02/01/2012).
Results of Phase I study:
In a phase I study a starting dose of 20 mg/m2 doxorubicin equivalents was chosen and 41 patients with advanced cancer disease were treated at dose levels of 20–340 mg/m2 doxorubicin equivalents . Treatment with INNO-206 was well tolerated up to 200 mg/m2 without manifestation of drug-related side effects which is a ~3-fold increase over the standard dose for doxorubicin (60 mg/kg). Myelosuppression and mucositis were the predominant adverse effects at dose levels of 260 mg/m2 and became dose-limiting at 340 mg/m2. 30 of 41 patients were assessable for analysis of response. Partial responses were observed in 3 patients (10%, small cell lung cancer, liposacoma and breast carcinoma). 15 patients (50%) showed a stable disease at different dose levels and 12 patients (40%) had evidence of tumor progression. (source: Invest New Drugs (2010) 28:14–19)
phase 2
CytRx Corporation (CYTR), a biopharmaceutical research and development company specializing in oncology, today announced that its oral presentation given by Sant P. Chawla, M.D., F.R.A.C.P., Director of the Sarcoma Oncology Center, titled “Randomized phase 2b trial comparing first-line treatment with aldoxorubicin versus doxorubicin in patients with advanced soft tissue sarcomas,” was featured in The Lancet Oncology in its July 2014 issue (Volume 15, Issue 8) in a review of the major presentations from the 2014 American Society of Clinical Oncology (ASCO) Annual Meeting.
“We are honored to have been included in The Lancet Oncology’s review of major presentations from ASCO and pleased that these important clinical findings are being recognized by one of the world’s premier oncology journals,” said Steven A. Kriegsman, CytRx President and CEO. “In clinical trials, aldoxorubicin has been shown to be a well-tolerated and efficacious single agent for the treatment of soft tissue sarcoma (STS) that lacks the cardiotoxicity associated with doxorubicin therapy, the current standard of care. We remain on track to report the full overall survival results from this trial prior to year-end 2014.”
The data presented at ASCO 2014 were updated results from CytRx’s ongoing multicenter, randomized, open-label global Phase 2b clinical trial investigating the efficacy and safety of aldoxorubicin compared with doxorubicin as first-line therapy in subjects with metastatic, locally advanced or unresectable STS. The updated trial results demonstrated that aldoxorubicin significantly increases progression-free survival (PFS), PFS at 6 months, overall response rate (ORR) and tumor shrinkage, compared to doxorubicin, the current standard-of-care, as a first-line treatment in patients with STS. The data trended in favor of aldoxorubicin for all of the major subtypes of STS
phase 3
Aldoxorubicin is currently being studied in a pivotal global Phase 3 clinical trial evaluating the efficacy and safety of aldoxorubicin as a second-line treatment for patients with STS under a Special Protocol Assessment with the FDA. CytRx is also conducting two Phase 2 clinical trials evaluating aldoxorubicin in patients with late-stage glioblastoma (GBM) and HIV-related Kaposi’s sarcoma and expects to start a phase 2b study in patients with relapsed small cell lung cancer
PATENTS WO 2000076551, WO 2008138646, WO 2011131314,
…………………….
WO 2014093815
http://www.google.com/patents/WO2014093815A1?cl=en
Anthracyclines are a class of antibiotics derived from certain types of Streptomyces bacteria. Anthracyclines are often used as cancer therapeutics and function in part as nucleic acid intercalating agents and inhibitors of the DNA repair enzyme topoisomerase II, thereby damaging nucleic acids in cancer cells, preventing the cells from replicating. One example of an anthracycline cancer therapeutic is doxorubicin, which is used to treat a variety of cancers including breast cancer, lung cancer, ovarian cancer, lymphoma, and leukemia. The 6-maleimidocaproyl hydrazone of doxorubicin (DOXO-EMCH) was originally synthesized to provide an acid-sensitive linker that could be used to prepare immunoconjugates of doxorubicin and monoclonal antibodies directed against tumor antigens (Willner et al., Bioconjugate Chem 4:521-527 (1993)). In this context, antibody disulfide bonds are reduced with dithiothreitol to form free thiol groups, which in turn react with the maleimide group of DOXO-EMCH to form a stable thioether bond. When administered, the doxorubicin-antibody conjugate is targeted to tumors containing the antigen recognized by the antibody. Following antigen-antibody binding, the conjugate is internalized within the tumor cell and transported to lysosomes. In the acidic lysosomal environment, doxorubicin is released from the conjugate intracellularly by hydrolysis of the acid-sensitive hydrazone linker. Upon release, the doxorubicin reaches the cell nucleus and is able to kill the tumor cell. For additional description of doxorubicin and
DOXO-EMCH see, for example, U.S. Patents 7,387,771 and 7,902,144 and U.S. Patent Application No. 12/619,161, each of which are incorporated in their entirety herein by reference.
[0003] A subsequent use of DOXO-EMCH was developed by reacting the molecule in vitro with the free thiol group (Cys-34) on human serum albumin (HSA) to form a stable thioether conjugate with this circulating protein (Kratz et al, J Med Chem 45:5523-5533 (2002)). Based on these results, it was
hypothesized that intravenously-administered DOXO-EMCH would rapidly conjugate to HSA in vivo and that this macromolecular conjugate would preferentially accumulate in tumors due to an “enhanced permeability and retention” (EPR) intratumor effect (Maeda et al., J Control Release 65:271-284 (2000)).
[0004] Acute and repeat-dose toxicology studies with DOXO-EMCH in mice, rats, and dogs identified no toxicity beyond that associated with doxorubicin, and showed that all three species had significantly higher tolerance for DOXO-EMCH compared to doxorubicin (Kratz et al, Hum Exp Toxicol 26: 19-35 (2007)). Based on the favorable toxicology profile and positive results from animal tumor models, a Phase 1 clinical trial of DOXO-EMCH was conducted in 41 advanced cancer patients (Unger et al, Clin Cancer Res 13:4858-4866 (2007)). This trial found DOXO-EMCH to be safe for clinical use. In some cases, DOXO-EMCH induced tumor regression.
[0005] Due to the sensitivity of the acid-cleavable linker in DOXO-EMCH, it is desirable to have formulations that are stable in long-term storage and during reconstitution (of, e.g., previously lyophilized compositions) and administration. DOXO-EMCH, when present in compositions, diluents and administration fluids used in current formulations, is stable only when kept at low temperatures. The need to maintain DOXO-EMCH at such temperatures presents a major problem in that it forces physicians to administer cold (4°C) DOXO-EMCH compositions to patients. Maintaining DOXO-EMCH at low temperatures complicates its administration in that it requires DOXO-EMCH to be kept at 4°C and diluted at 4°C to prevent degradation that would render it unsuitable for patient use. Further, administration at 4°C can be harmful to patients whose body temperature is significantly higher (37°C).
[0006] Lyophilization has been used to provide a stable formulation for many drugs. However, reconstitution of lyophilized DOXO-EMCH in a liquid that does not maintain stability at room temperature can result in rapid decomposition of DOXO-EMCH. Use of an inappropriate diluent to produce an injectable composition of DOXO-EMCH can lead to decreased stability and/or solubility. This decreased stability manifests itself in the cleavage of the linker between the doxorubicin and EMCH moieties, resulting in degradation of the DOXO-EMCH into two components: doxorubicin and linker-maleimide. Thus, stable,
reconstituted lyophilized solutions of anthracycline-EMCH (e.g., DOXO-EMCH), and injectable compositions containing the same, are required to solve these problems and to provide a suitable administration vehicle that can be used reasonably in treating patients both for clinical trials and commercially.
DOXO-EMCH. The term “DOXO-EMCH,” alone or in combination with any other term, refers to a compound as depicted by the following structure:
OH
DOXO-EMCH is also referred to as (E)-N’-(l-((2S,4S)-4-(4-amino-5-hydroxy-6- methyl-tetrahydro-2H-pyran-2-yloxy-2,5 , 12-trihydroxy-7-methoxy-6, 11- dioxol,2,3,4,6,l l-hexahydrotetracen-2-yl)-2-hydroxyethylidene)-6-(2,5-dioxo-2H- pyrrol- 1 (5H)yl)hexanehydrazide»HCl.
………………………………
CN 102675385
http://www.google.com/patents/CN102675385A?cl=en
According to literature reports, (eg see David Willner et al, “(6_Maleimidocaproyl) hydrazoneof Doxorubicm-A New Derivative for the Preparation ofImmunoconjugates oiDoxorubicin,” Bioconjugate Chem. 1993,4, 521-527; JK Tota Hill, etc. man, “The method of preparation of thioether compounds noir,” CN1109886A, etc.), adriamycin 13 – bit hydrazone derivative synthesis and the main process are as follows:
[0004]
[0005] First, maleic anhydride and 6 – aminocaproic acid was refluxed in a large number of acid reaction ko ni acid I; agent under the action of the ring after the cyclization maleimidocaproic acid 2 (yield 30-40% ), cyclic acid anhydride mixture is generally ko, trimethyl silyl chloride and tri-amines such ko; maleimido aminocaproic acid tert-butyl ester with hydrazine to condensation to give 2 – (6 – aminocaproic maleimido ) hydrazine carboxylic acid tert-butyl ester 3 (yield 70-85%), the condensing agent is N-methylmorpholine and isobutyl chloroformate; 3 in a large number of trifluoroacetic acid deprotection ko maleimido ko has trifluoroacetic acid hydrazide 4 (yield 70%); the doxorubicin hydrochloride salt with a ko in trifluoroacetic acid catalyzed condensation in methanol solvent to doxorubicin hydrazone product was obtained (yield 80%) .
[0006] The synthetic method the yield is low (in particular, by maleic acid imido step 2), the total yield of not more than 20%, and the solvent consumption is large, adriamycin hydrazone product per Malek consumes about ko acid reaction solvent, 70mL, tetrahydrofuran 300mL, ko trifluoroacetic acid 40mL, and because the 2 – (6 – maleimido hexanoyl)-hydrazine carboxylic acid tert-butyl ester was purified by column chromatography required, but also to consume a large amount of Solvent. This has resulted in synthesis post-processing complex process, complicated operation. And because the end product of the synthesis of doxorubicin hydrazone ko using trifluoroacetic acid, inevitably there will be in the product ko trifluoroacetic acid impurities, not divisible. Based on the high cost of such a route exists, yield and production efficiency is low, Eri Arts route operational complexity and other shortcomings, is obviously not suitable for mass production, it is necessary to carry out improvements or exploring other Eri Arts synthesis methods.
doxorubicin hydrazone derivative,
Wherein n is an integer of 1-15, characterized in that said method comprises the steps of: (1) the maleic acid chloride of the formula H2N-(CH2) n-COOH amino acid I b in the presence of a base prepared by condensation of maleimido group steps I c acid,
(2) maleic acid imido group I c and then with an acylating reagent of tert-butyl carbazate in the presence of a base in the reaction of step I d,
(3) I d deprotection with trifluoroacetic acid, the alkali and removing trifluoroacetic acid to obtain the maleimido group I e hydrazide steps
(4) an imido group of maleic hydrazide I e and doxorubicin hydrochloride catalyzed condensation of hydrogen chloride to obtain a final product hydrazone derivative of doxorubicin,
[0028]
[0049] The butene-ni chloride 15. 2g (0. Imol) was dissolved in 25mL of chloroform was dried by adding anhydrous potassium carbonate 27. 6g (0. 2mol), the gas and gas protection and conditions of 0 ° C was added dropwise 6 – aminocaproic acid 13. 2g (0. ImoI) in chloroform (50mL) solution, add after reaction at room temperature for 3 hours. Washed with saturated brine, dried over anhydrous magnesium sulfate, suction filtered, concentrated under reduced pressure. The residue was recrystallized from alcohol ko maleimido acid (Compound c) 18. 8g, 90% yield, m.p. :85-87 ° C.
[0050] Compound c 10. 5g (50mmol) and thionyl chloride crab 5. 3mL (75mmol) was heated under reflux the mixture I. 5 hours and concentrated under reduced pressure in an argon atmosphere under the conditions of 0 ° C and added dropwise to the hydrazine carboxylic acid tert-butyl ester 6.6g (50mmol) amine with a three ko
10. 8mL (75mmol) in anhydrous ko ether (50mL) solution added after the reaction was continued at room temperature for I. 5 hours. Washed with 5% hydrochloric acid, 5% sodium bicarbonate, and saturated brine, dried over anhydrous magnesium sulfate overnight, filtered with suction to give the compound of d ko ether solution. The solution was cooled to 0 ° C, was added dropwise trifluoroacetic acid ko 7. 4mL (100mmOl), After the addition the reaction was continued for 10 minutes, suction filtered, the filter cake was washed twice with ether, ko and dried in vacuo to give 6 – maleic acid sub-aminocaproic acid hydrazide trifluoro-ko 12. 2g, 72% yield, m.p. 99-102 ° C. IOmL this salt is added to sodium hydroxide (10%) solution, stirred for a while, with ko extracted with ether, the organic layer was washed with water, dried over anhydrous magnesium sulfate, and concentrated to give 6 – aminocaproic maleimido hydrazide (compound e) 7. Sg, 70% yield.
[0051] The doxorubicin hydrochloride 0. 58g (Immol) with compound e 0. 45g (2mmol) was dissolved in 150mL of anhydrous methanol, passing about 2mmol of dry hydrogen chloride, under argon, at room temperature protected from light and reaction conditions 24 inches. Concentrated under reduced pressure at room temperature, the solid was washed with about IOOmL ko nitrile, and dried in vacuo doxorubicin 6 – aminocaproic maleimido hydrazone O. 63g, 80% yield. 1H NMR (CD3OD) δ: 7. 94 (bd, 1H), 7. 82 (t, 1H), 7. 55 (d, 1H), 6. 78 (s, 2H), 5. 48 (s, 1H ), 5. 07 (t, 1H), 4 · 59 (d, 1H), 4 · 21 (m, 1Η), 4 · 02 (s, 3H), 3 · 63-3. 30 (m, 5H) , 2 · 55-2. 26 (m, 4H), 2. 19-1. 88 (m, 3Η), I. 69-1. 18 (m, 12Η, I. 26). [0052] Although specific reference to the above embodiments of the present invention will be described, it will be understood that in the appended claims without departing from the invention as defined by the spirit and scope of the skilled person can be variously truncated, substitutions and changes. Accordingly, the present invention encompasses these deletions, substitutions and changes.
………………………………….
US 5622929
http://www.google.co.in/patents/US5622929
http://www.google.co.in/patents/EP0554708A1
Method A:
As noted below, Method A is the preferred method when the Michael Addition Receptor is a maleimido moiety.
Alternatively, the Formula (IIa) compound may be prepared by reaction of the drug with a hydrazide to form an intermediate hydrazone drug derivative followed by reaction of this compound with a Michael Addition Receptor containing moiety according to the general process described in Method B:
…………………………………….
http://www.google.co.in/patents/WO2012167255A1?cl=en
Synthesis of DOXO-EMCH
The synthesis of DOXO-EMCH was done initially in accordance with that previously published by Willner and co-workers (Bioconjugate Chem., 4:521-527, 1993). Problems arose in the initial addition of the 6-maleimidocaproylhydrazine to the C-13 ketone of doxorubicin. HPLC results did not give a good yield of product, only 50-60%. Upon further analysis, we determined TFA was not needed to catalyze the reaction, and instead used pyridine. With pyridine, chromatograms from the HPLC showed 90% DOXO-EMCH relative to 10% DOX. The pyridine may have improved the yield by serving as a base to facilitate formation of the hydrazone. Another problem we encountered in the synthesis was purification of the final product. According to Willner’ s method, 5 volumes of acetonitrile (ACN) were to be added to a concentrated methanolic solution of crude DOXO-EMCH to achieve crystallization after 48 h at 4 °C. By this method, only 10-20%) of the desired product precipitated. To obtain a better yield, the crystallization step was done 4 times with 6 volumes of ACN used in each step. A lesser amount of methanol was needed each time, as less product remained in solution. Even with the multiple crystallizations, a final yield of only 59% was obtained. Various other methods for crystallization were explored, including using different solvents and increasing the initial solubility in methanol by heat, but none gave better results. 1.2 Rate of Hydrolysis of DOXO-EMCH at Varying pH
Subsequent pH studies to determine the rate of hydrolysis of the hydrazone were carried out as a benchmark for later hydrolysis experiments with PPD-EMCH. The results of the hydrolysis experiments showed that at lower pH, the hydrolysis reaction proceeded very quickly in the formation of DOX. Moreover, at higher pH the hydrazone proved to be very robust in that its degradation is very slow.
General HPLC instruments and methods
Analytical HPLC methods were performed using a Hewlett-Packard/ Aligent 1050/1100 chromatograph with an auto injector, diode array UV-vis absorption detector. Method 1.1 : Analytical HPLC injections were onto an Aligent Zorbax Eclipse XDB-C18 reversed phase column, 4.6 mm x 150 mm, eluting at 1.0 mL/min. A gradient of acetonitrile/20 mM sodium phosphate buffer (pH 6.9), 80% buffer to 55% at 10 min, 55% to 40% at 12 min, 40% to 80% at 13 min. Retention times: at 480 nm, DOX (9.4 min), DOXO-EMCH (1 1.2 min).
Synthesis of DOXO-EMCH
The synthesis of DOXO-EMCH was accomplished using the procedure reported by Willner et al, with several changes to improve the yield (Willner, D., et al.,
Bioconjugate Chem., 4:521-27, 1993). DOX’HCl (20 mg, 34 μιηοΐ) was dissolved in 6 mL of methanol. Pyridine (12.53 μί) was added to the solution, followed by 35.4 mg
EMCH’TFA. The reaction was stirred at room temperature overnight. By HPLC, the reaction was 90% complete. The solvent was evaporated to dryness by rotary evaporation. A minimal amount of methanol was used to dissolve the solid, and six volumes of acetonitrile at 4 °C were added to the solution. The resulting solution was allowed to sit undisturbed at 4 °C for 48 h for crystallization. The precipitate was collected, and the crystallization method was repeated 4 times. The resulting solids were combined and washed three times with 1 : 10 methanokacetonitrile. The final yield of DOXO-EMCH was 11.59 mg, 58%. HPLC Method 1.1 was used. NMR spectra corresponded to those previously given by Willner (Bioconjugate Chem. 4:521-27. 1993).
…………………………….
http://www.google.co.in/patents/US20070219351
DOXO-EMCH, the structural formula of which is shown below,
…………………………………
SEE
(6-Maleimidocaproyl)hydrazone of doxorubicin – A new derivative for the preparation of immunoconjugates of doxorubicin
Bioconjugate Chem 1993, 4(6): 521
| References |
1: Kratz F, Azab S, Zeisig R, Fichtner I, Warnecke A. Evaluation of combination therapy schedules of doxorubicin and an acid-sensitive albumin-binding prodrug of doxorubicin in the MIA PaCa-2 pancreatic xenograft model. Int J Pharm. 2013 Jan 30;441(1-2):499-506. doi: 10.1016/j.ijpharm.2012.11.003. Epub 2012 Nov 10. PubMed PMID: 23149257.
2: Walker L, Perkins E, Kratz F, Raucher D. Cell penetrating peptides fused to a thermally targeted biopolymer drug carrier improve the delivery and antitumor efficacy of an acid-sensitive doxorubicin derivative. Int J Pharm. 2012 Oct 15;436(1-2):825-32. doi: 10.1016/j.ijpharm.2012.07.043. Epub 2012 Jul 28. PubMed PMID: 22850291; PubMed Central PMCID: PMC3465682.
3: Kratz F, Warnecke A. Finding the optimal balance: challenges of improving conventional cancer chemotherapy using suitable combinations with nano-sized drug delivery systems. J Control Release. 2012 Dec 10;164(2):221-35. doi: 10.1016/j.jconrel.2012.05.045. Epub 2012 Jun 13. PubMed PMID: 22705248.
4: Sanchez E, Li M, Wang C, Nichols CM, Li J, Chen H, Berenson JR. Anti-myeloma effects of the novel anthracycline derivative INNO-206. Clin Cancer Res. 2012 Jul 15;18(14):3856-67. doi: 10.1158/1078-0432.CCR-11-3130. Epub 2012 May 22. PubMed PMID: 22619306.
5: Kratz F, Elsadek B. Clinical impact of serum proteins on drug delivery. J Control Release. 2012 Jul 20;161(2):429-45. doi: 10.1016/j.jconrel.2011.11.028. Epub 2011 Dec 1. Review. PubMed PMID: 22155554.
6: Elsadek B, Kratz F. Impact of albumin on drug delivery–new applications on the horizon. J Control Release. 2012 Jan 10;157(1):4-28. doi: 10.1016/j.jconrel.2011.09.069. Epub 2011 Sep 16. Review. PubMed PMID: 21959118.
7: Kratz F, Fichtner I, Graeser R. Combination therapy with the albumin-binding prodrug of doxorubicin (INNO-206) and doxorubicin achieves complete remissions and improves tolerability in an ovarian A2780 xenograft model. Invest New Drugs. 2012 Aug;30(4):1743-9. doi: 10.1007/s10637-011-9686-5. Epub 2011 May 18. PubMed PMID: 21590366.
8: Boga C, Fiume L, Baglioni M, Bertucci C, Farina C, Kratz F, Manerba M, Naldi M, Di Stefano G. Characterisation of the conjugate of the (6-maleimidocaproyl)hydrazone derivative of doxorubicin with lactosaminated human albumin by 13C NMR spectroscopy. Eur J Pharm Sci. 2009 Oct 8;38(3):262-9. doi: 10.1016/j.ejps.2009.08.001. Epub 2009 Aug 18. PubMed PMID: 19695327.
9: Graeser R, Esser N, Unger H, Fichtner I, Zhu A, Unger C, Kratz F. INNO-206, the (6-maleimidocaproyl hydrazone derivative of doxorubicin), shows superior antitumor efficacy compared to doxorubicin in different tumor xenograft models and in an orthotopic pancreas carcinoma model. Invest New Drugs. 2010 Feb;28(1):14-9. doi: 10.1007/s10637-008-9208-2. Epub 2009 Jan 8. PubMed PMID: 19148580.
10: Kratz F. Albumin as a drug carrier: design of prodrugs, drug conjugates and nanoparticles. J Control Release. 2008 Dec 18;132(3):171-83. doi: 10.1016/j.jconrel.2008.05.010. Epub 2008 May 17. Review. PubMed PMID: 18582981.
11: Unger C, Häring B, Medinger M, Drevs J, Steinbild S, Kratz F, Mross K. Phase I and pharmacokinetic study of the (6-maleimidocaproyl)hydrazone derivative of doxorubicin. Clin Cancer Res. 2007 Aug 15;13(16):4858-66. PubMed PMID: 17699865.
12: Lebrecht D, Walker UA. Role of mtDNA lesions in anthracycline cardiotoxicity. Cardiovasc Toxicol. 2007;7(2):108-13. Review. PubMed PMID: 17652814.
13: Kratz F. DOXO-EMCH (INNO-206): the first albumin-binding prodrug of doxorubicin to enter clinical trials. Expert Opin Investig Drugs. 2007 Jun;16(6):855-66. Review. PubMed PMID: 17501697.
14: Kratz F, Ehling G, Kauffmann HM, Unger C. Acute and repeat-dose toxicity studies of the (6-maleimidocaproyl)hydrazone derivative of doxorubicin (DOXO-EMCH), an albumin-binding prodrug of the anticancer agent doxorubicin. Hum Exp Toxicol. 2007 Jan;26(1):19-35. PubMed PMID: 17334177.
15: Lebrecht D, Geist A, Ketelsen UP, Haberstroh J, Setzer B, Kratz F, Walker UA. The 6-maleimidocaproyl hydrazone derivative of doxorubicin (DOXO-EMCH) is superior to free doxorubicin with respect to cardiotoxicity and mitochondrial damage. Int J Cancer. 2007 Feb 15;120(4):927-34. PubMed PMID: 17131338.
16: Di Stefano G, Lanza M, Kratz F, Merina L, Fiume L. A novel method for coupling doxorubicin to lactosaminated human albumin by an acid sensitive hydrazone bond: synthesis, characterization and preliminary biological properties of the conjugate. Eur J Pharm Sci. 2004 Dec;23(4-5):393-7. PubMed PMID: 15567293.
| EP0169111A1 * | Jun 18, 1985 | Jan 22, 1986 | Sanofi | Cytotoxic conjugates useful in therapy, and process for obtaining them |
| EP0269188A2 * | Jun 18, 1985 | Jun 1, 1988 | Elf Sanofi | Cytotoxic conjugates useful in therapy, and process for obtaining them |
| EP0306943A2 * | Sep 8, 1988 | Mar 15, 1989 | Neorx Corporation | Immunconjugates joined by thioether bonds having reduced toxicity and improved selectivity |
| EP0328147A2 * | Feb 10, 1989 | Aug 16, 1989 | Bristol-Myers Squibb Company | Anthracycline immunoconjugates having a novel linker and methods for their production |
| EP0398305A2 * | May 16, 1990 | Nov 22, 1990 | Bristol-Myers Squibb Company | Anthracycline conjugates having a novel linker and methods for their production |
| EP0457250A2 * | May 13, 1991 | Nov 21, 1991 | Bristol-Myers Squibb Company | Novel bifunctional linking compounds, conjugates and methods for their production |
Aldoxorubicin, CytRx, CANCER, INNO-206, PHASE 3, oncology, Soft Tissue Sarcoma